The Early Development of the GI Tract and the Immune System
Thomas A. Kruzel, ND
Breast feeding and the introduction of solid foods during infancy have long been recognized by naturopathic physicians as being vital to the development of children’s immune systems, which ultimately translates to good health and well being throughout their lives. Much has been written about the quality of foods and availability of nutrients needed for growth, development, and maintenance of health.1 Naturopathic medicine, with its emphases on diet and nutrition, has long recognized that the early development of the gastrointestinal tract and its relationship to development of immunity sets the stage for how individuals deal with health challenges throughout the rest of their lives. Certainly, this is all the more important in light of recent findings of high levels of environmental toxins, increasing resistance of microorganisms to antibiotics, and ever-changing viral organisms that we will experience throughout our existences. Additionally, the added pressures of nutrient deplete and higher lectin-containing foods produced by corporate farming methods and genetically modified foods upon the gastrointestinal tract are only recently being recognized.
The benefits of breast feeding and its effects on immune system development have been extensively reviewed and will not be covered here. An excellent review article can be found on this subject in the recent Natural Medicine Journal.2 Breast feeding has a significant effect on the growth, development, and function of the epithelium and immune and nervous systems of the gastrointestinal tract and is an integral component of the infant’s innate defense system. Properties in breast milk, such as cytokines, lactoferrin, glycoconjugates, oligosaccharides, white blood cells, and immunoglobulins, help protect the developing infant’s GI tract from colonization by bacteria associated with necrotizing enterocolitis, as well as allergens. Antibodies found in breast milk have been shown to reflect the antigenic repertoire of the mother’s gastrointestinal and respiratory tracts. It has also been shown that while there is variation from mother to mother in the types of antibodies conveyed through breast milk, if the bacterial organisms are similar in both environments of mother and infant, then a greater specificity is achieved.3 All of these factors help to convey a selective advantage to beneficial organisms, such as bifidobacteria and lactobacilli, needed for GI tract and immune development in the infant. Additionally, the volume of the thymus has been found to be greater in breastfed infants than non-breastfed infants, which lends credence to the GI/immune system development connection.4[protected]
Breast feeding also has a positive effect on developmentally delayed or premature infants while providing protection to an immature GI mucosa. Breast milk helps the GI tract make the transition from an intrauterine environment to an extrauterine environment that exposes the infant to considerably more toxic substances. Breast feeding also provides anti-inflammatory properties to the developing GI tract, as well as enzymes, to compensate while the pancreas and growth factors that aid in establishment of normal gut flora develop.5
The developing gastrointestinal tract is essentially sterile at birth and subsequently colonized by microbe s acquired from the mother and the surrounding environment. Children delivered vaginally acquire microorganisms present in the vaginal tract (primarily lactobacilli), while children delivered by Cesarean section (C-section) acquire organisms present on the skin and surrounding environment. The surrounding environment is most often a hospital setting which fosters its own population of microorganisms not normally found in the community at large. In each case, the types of bacteria are different, with children being delivered by C-section experiencing a greater risk of developing pathogenic intestinal flora. Additionally, a delay in colonization of the developing GI tract by protective bacteria also places the infant at risk for the development of a limited number of species, thus increasing risk for development of necrotizing enterocolitis.6 Bacterial diversity early on plays a significant role in preparing the immune system to be able to respond to diseases throughout the individual’s life.
s acquired from the mother and the surrounding environment. Children delivered vaginally acquire microorganisms present in the vaginal tract (primarily lactobacilli), while children delivered by Cesarean section (C-section) acquire organisms present on the skin and surrounding environment. The surrounding environment is most often a hospital setting which fosters its own population of microorganisms not normally found in the community at large. In each case, the types of bacteria are different, with children being delivered by C-section experiencing a greater risk of developing pathogenic intestinal flora. Additionally, a delay in colonization of the developing GI tract by protective bacteria also places the infant at risk for the development of a limited number of species, thus increasing risk for development of necrotizing enterocolitis.6 Bacterial diversity early on plays a significant role in preparing the immune system to be able to respond to diseases throughout the individual’s life.
As well as mode of delivery, the developing gastrointestinal ecosystem is also influenced by dietary and environmental conditions. Along with the introduction of micro-flora, development of the gastrointestinal system is influenced by four major determinants: genetic makeup, intrinsic developmental and biological clock, endogenous regulatory mechanisms, and environmental influences.7 Secretory and absorptive functions of the GI tract, while present at 26 weeks of gestation, are incomplete and still developing at birth. Additionally, pancreatic and gastric enzyme secretion do not begin to fully develop until after birth and are influenced by diet, development of cholecystokinin, gastrin, secretin, insulin, insulin-like growth factor, epidermal growth factor, and thyroid function.
In the developing gastrointestinal tract, the tissues associated with digestion and absorption do not develop simultaneously. Rather, morphologic and functional development progress at a rate unique to the individual and are influenced by mechanical function of the GI tract. The major determinants of this readiness include suck–swallow coordination, gastroesophageal sphincter tone, gastric emptying, and intestinal motility. Additionally, malnutrition, improper nutrition, or exogenous nutrition from formula feeding may restrict morphologic and biochemical development of the GI tract. This is especially problematic in premature infants, particularly those who need parenteral feeding while in the hospital.8
Some authors9 break down the development of gastrointestinal flora into 4 phases, with the first being an acquisitional phase during the first 1 to 2 weeks of life; the second being the period of breast feeding for growth and development; the third being the introduction of solid foods and the subsequent decrease in breast feeding; and the fourth being the conversion to an adult-like diet pattern. Feeding patterns may also play a role in developing intestinal flora in that infants usually feed on demand within the first few weeks of life, followed by more of a schedule as breast milk increases. The first few days of breast feeding are taken up by the removal of colostrum to be replaced by larger amounts of breast milk. Colostrum provides active immunity as the infant’s GI tract begins to establish its flora.10
A breast-fed, full-term infant has a preferred intestine micro-flora in which bifidobacteria, lactic acid bacteria, and enterobacteria predominate over potentially harmful bacteria, such as E. coli, streptococci, bacteroides, and clostridia, whereas in formula-fed infants, coliforms, enterococci, and bacteroides predominate.11 The gastrointestinal tracts of children delivered by C-section are eventually colonized with similar organisms to those found in vaginally delivered infants if the infant is subsequently breast-fed. What has also been noted is that breast-fed infants, when formula is added, begin to develop a micro-flora similar to formula-fed infants. Whether this contributes to later development of allergies, as is seen in predominantly formula-fed infants, remains to be seen. In my experience, children who breast feed for at least the first year, even if it is supplemented with formula, are less likely to develop allergies later on. In both groups, with the introduction of solid foods, obligate anaerobes increase in number and diversity until a pattern similar to that in adult humans is achieved by about 2 years of age.
The presence of large numbers of lactobacilli inhabiting the proximal intestinal tract correlates with the presence of stratified squamous epithelium and the ability of lactobacillus cells to adhere to the epithelium. This area of the GI tract has relatively few microorganisms compared to the distal small intestine and the colon, as bacterial flora continue to proliferate as they act to digest foods. Conversely, mucus production begins in earnest in the duodenum to provide a protective barrier as we ll as a binding agent for fecal matter and as a protection against the burgeoning bacterial population.
ll as a binding agent for fecal matter and as a protection against the burgeoning bacterial population.
When solid foods begin to be introduced, bacterial profiles obtained from stool cultures begin to change so that breast-fed infants show patterns similar to formula-fed infants. This pattern continues to change with the increasing complexity of foods as the infant matures and he or she is better able to digest and assimilate them. Therefore, a more complex food introduced before the infant’s GI tract is mature enough to handle it places him or her at risk for developing an altered flora that increases his or her risk for development of allergies, as well as necrotizing enetercolitis.
Intrinsic or host-related factors that influence microbial succession include genetic makeup, individual physiology, nutrient intake, pH, microbe interaction/competition, and physiologic factors such as gut motility, internal secretions, bile acids, and immune response. Additionally, defense mechanisms such as mucus secretion, stool pattern, and whether the immune system has been overtaxed by immunizations or antibiotic use play a role.
Generally, fluctuations in fecal microbial populations are greater for infants than adults with larger day-to-day and diet-induced variations, based upon stool culture analysis. However, stool culture only determines aerobic bacteria that colonize the large intestine, forgoing anaerobic bacterial species, which are the predominant populations. Polymerase chain reaction (PCR) studies of stool samples from infants that evaluate anaerobic bacterial populations show considerable diversity within the first 6 months of life, gradually developing adult patterns as they mature.12 The authors note that colonization occurs due to common environmental exposures during birth. What must be considered as well is that microorganisms are also obtained as children explore their worlds through oral and tactile methods while playing on the floor, in the dirt, or with other children. While initially the gut bacteria demonstrate fluctuations as the various micro-biota attempt to establish predominance, eventually more stable, adult-like patterns emerge due to changes in the gut environment and mucosa with maturation. The diversity and variation found thus reflect the corresponding individuality of these exposures. As a whole, the gastrointestinal micro-biota can be considered a metabolically adaptable and rapidly renewable organ of the body. It is its own intrinsic ecosystem that will live in a symbiotic relationship with us throughout our lives.
In older children and adults, the intestinal protective barrier is composed of mucin and secretory IgA, which is the primary line of defense against viruses, bacteria, parasites, and food antigens. Secretory IgA is involved in nonspecific inflammatory reactions via the alternative complement pathway. Mucin is high in glycoproteins, which have similar glycoprotein structures to the ABO and the MN blood group systems. As part of the mechanism of host defense, sialogen content (sialic acid) increases the affinity of the complement enzyme C3 for its control factor substance H. Sialic acid has a greater affinity for viral and bacterial proteins, thus providing a barrier against invasion. Therefore, a high sialic acid content translates to better control of complement cascade, as seen with secretors, while a low sialic acid content translates to a poorer control of complement cascade, resulting in greater and less modulated inflammation, as is seen with non-secretors.13 While this difference has a greater significance in older children and adults, infants have a different mucin/sialogen content, which conveys greater protection to the developing GI system, and therefore we almost never see the severe inflammatory reactions to offending foods during this period. If they occur, it is probably due to abnormal flora which utilize glycoproteins as food when the mucus protective barrier has broken down.
Antibiotic therapy and other drug therapies also have a major effect on succession of the bacterial flora of the gastrointestinal tract. Antimicrobial agents can have specific effects on individual populations of the micro-flora, rather than a general, nonspecific suppression of all microbes. Secondly, the remaining microbial profile will influence the populations that emerge after treatment has stopped. Lastly, the effects of antibiotic therapy can persist for some time afterward, often for years. Because antibiotics have specific modes of action and organisms exhibit differing sensitivities, it is not surprising that particular antibiotics influence individual populations, thus skewing the intestinal flora while conveying a selective advantage to another species.
Once disruption of normal micro-biota balance occurs because of frequent antibiotic use, pathogenic bacteria have the ability to not only produce bacteriocidins in order to gain a selective advantage over other bacteria, but proteases, ureases, and other substances which alter intestinal defenses and affect host immunity. Additionally, their propensity to contribute to bowel toxemia through production of indole, skatole, phenol, and hydrogen sulfide is well-documented.14 Therefore, prolonged or even periodic use of antibiotics can affect the bacterial ecosystem to some extent and needs to be corrected as soon as possible before the change becomes fixed.15
The gastrointestinal mucosal barrier must be able to provide for digestion and absorption of nutrients while not provoking an immune response to food substances and existing with the developing micro-flora. One of the keys to obtaining this dynamic is the early development of the floral balance, which imparts an intricate balance between GI bacteria, digestion, protection against microbes, assimilation of nutrients, and development of gut associated lymphoid tissue (GALT). Colonization with a diverse bacterial flora is necessary for the secretion of IgA and the development of T and B cell populations. Once this is established, a recognition pattern (Toll-like receptors) on gut epithelial and lymphoid cells acts to mediate immune response to bacterial molecular patterns, which conveys immunity.16
Some authors16 have recognized that a lack of bacterial diversity early on, either through paucity of contact, public health measures to decrease exposures, or frequent antibiotic use, results in an increase in atopic and autoimmune diseases simply because the infant’s immune system has not been allowed to develop properly. Along these lines, in a study of children raised on an anthroposophic diet, comprising vegetables spontaneously fermented by lactobacilli and the restricted use of antibiotics, anti-pyretics, and vaccinations, it was concluded that the lower incidence of atopic diseases found in these families was due to the lifestyle’s contributions to development of the children’s bacterial flora.17 A similar recognition by naturopathic physicians of the propensity to develop chronic degenerative and autoimmune diseases has been extensively documented in writings by the profession for over 100 years.
It is not unusual in clinical practice to see children who have difficulty assimilating any number of infant formulas. Many of these have lactose or high fructose corn syrup as a main ingredient, which provide calories but little more. Additionally, many of them contain whey protein and soy oil. Some of the most common causes of food allergies in infants and young children are seen when milk and soy are predominant parts of their diet. The hydrolyzed starches in many infant formulas will alter the micro-flora balance and can contribute to hyperosmolar-induced diarrhea.18
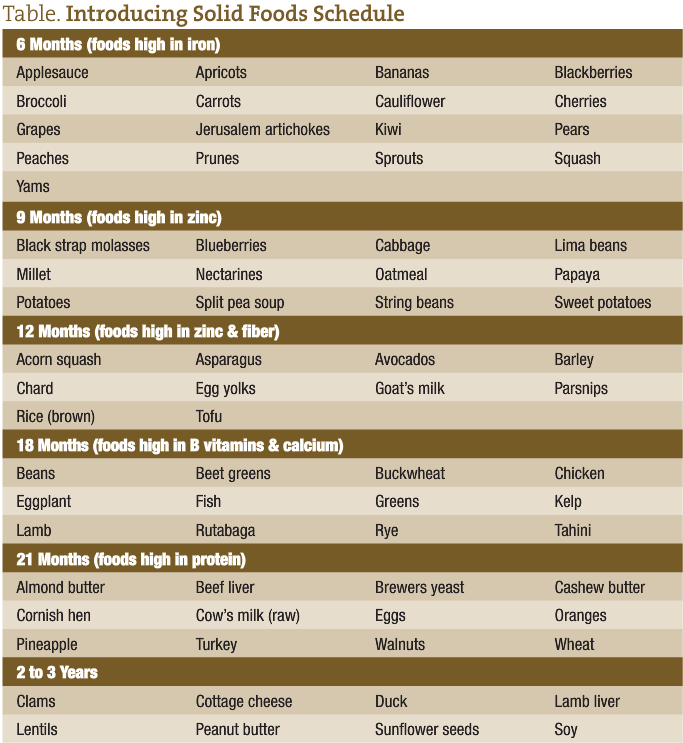
Introduction of solid foods usually begins at about 5 to 6 months of age, or about the time that the child is teething. Concurrently, the infant is grasping things and exploring his or her environment by touch and taste. The foods listed on the schedule provided are in order of increasing complexity and lectin content. This is important in that while the infant has derived much of his or her immunity from breast milk, his or her immune system must learn to stand on its own. If the infant is subjected to foods of greater complexity than his or her developing immune system can handle, conditions such as chronic diarrhea and/or constipation ensue. Colic may also be a sign that his or her gastrointestinal tract isn’t quite ready for what it is being fed.
Most of the foods that children begin with at 6 months are colored, soft, sweet, high in antioxidants, and provide stimulation for the formation of antibodies and development of GALT. They are also high in iron, an essential nutrient for the continued development of the GI mucosa and growth. These foods are also high in polyphenols, which alter the growth of microflora and adhesion to the mucus layer. Naringenin (a bioflavinoid derived from foods such as oranges and grapefruit) and quercitin (from capers, apples, green tea, onions, red grapes, citrus, broccoli, cherries, raspberries, cranberries, prickly pear fruit, and honey) act to support the growth of lactobacillus while inhibiting pathogenic organisms such as salmonella.15
At 9 months, foods higher in fiber and zinc are introduced. Parents often notice a change in stool pattern, color, consistency, and frequency when these foods are introduced. By now, the bacterial flora are undergoing a transformation because of dietary changes as more anaerobic bacteria begin to colonize the GI tract.
At 12 months, foods higher in protein and fats, as well as fiber, can be introduced. While goat’s milk is listed here, it can be introduced earlier if for some reason breast feeding is not available, rather than using prepackaged infant formula. A formula for goat’s milk that I have found effective accompanies this article, but there are many variations on the theme.
At 18 months, more complex foods requiring greater levels of digestive enzymes to break them down in order to aid in assimilation are found. By this time, the child’s salivary production, pancreas, liver, and gastrointestinal tract have developed the enzyme systems needed to accomplish this task. These foods are higher in protein, B vitamins, and minerals such as calcium and magnesium.
At 21 months, more complex foods are introduced that are higher in protein, as well as other nutrients needed for the now rapid growth that is occurring. Some of these foods, such as clams, lentils, sunflower seeds, and wheat, are high in lectins, which continue to develop the GALT.
The increasing complexity of foods introduced in an incremental and ordered manner provides the basis for the continued development of the child’s immune system. But what is also needed in order for the immune system to develop is continued exposure to the microorganisms that make it strong. Therefore, children who play in the dirt develop better immune systems because of the bacterial diversity that allows the developing immune system to identify the bad guys early on, when optimal development is taking place, rather than later in life, when the thymus and gut-associated lymphatic tissue are not as primed for development. Additionally, that a child has periodic fevers and that they be allowed to do their work without the use of antipyretics or antibiotics really provides a boost to immune function and development. Children will also have periodic loose stools and diarrhea as their GI tracts sort out all of the competing microorganisms vying for their places in the developing ecosystem. These run-of-the-mill variety loose stools are self-limiting and provide a protective mechanism against an over growth of abnormal bacteria.
Naturopathic medicine has always viewed the human organism as living in a symbiotic and harmonious relationship with the external environment. This involves not only the micro-biota that inhabit our gastrointestinal tracts but the foods we eat, the environment in which we live, and the state of health of the planet. Certainly, the development of our immunity early in life through the introduction of solid foods, the development of good dietary habits and patterns, and the recognition that our immune systems need training rather than suppression early on will translate to healthier individuals throughout our lives.
 Thomas A. Kruzel, ND is a naturopathic physician who is in private practice at the Scottsdale Natural Medicine & Healing Clinic in Scottsdale, AZ. He is the former Vice President of Clinical Affairs and the Chief Medical Officer at the Southwest College of Naturopathic Medicine in Phoenix, AZ. He received a BA in Biology from the California State University at Northridge and his Doctorate of Naturopathic Medicine degree from the National College of Naturopathic Medicine. He has been an Associate Professor of Medicine at National College of Naturopathic Medicine. He is the author of the Homeopathic Emergency Guide A Quick Reference Handbook to Effective Homeopathic Care. He is Past President of the American Association of Naturopathic Physicians and was selected as Physician of the Year by the AANP in 2000 and Physician of the Year by the Arizona Naturopathic Medical Association in 2003.
Thomas A. Kruzel, ND is a naturopathic physician who is in private practice at the Scottsdale Natural Medicine & Healing Clinic in Scottsdale, AZ. He is the former Vice President of Clinical Affairs and the Chief Medical Officer at the Southwest College of Naturopathic Medicine in Phoenix, AZ. He received a BA in Biology from the California State University at Northridge and his Doctorate of Naturopathic Medicine degree from the National College of Naturopathic Medicine. He has been an Associate Professor of Medicine at National College of Naturopathic Medicine. He is the author of the Homeopathic Emergency Guide A Quick Reference Handbook to Effective Homeopathic Care. He is Past President of the American Association of Naturopathic Physicians and was selected as Physician of the Year by the AANP in 2000 and Physician of the Year by the Arizona Naturopathic Medical Association in 2003.
(3-References): 1. Schmid R. Traditional Foods Are Your Best Medicine. Stratford, CT: Ocean View Publications; 1987.
2. Clinton C. Development of the Infant Immune Function and the Effects of Breast Milk. Natural Medicine Journal. August 2010;2(8).
3. Neu J, Douglas-Escobar M. Gastrointestinal Development: Implications for Infant Feeding. Nutrition in Pediatrics. 4th ed. Hamilton, ON: BC Decker Inc; 2008.
4. Hasselbalch H, Jeppesen DL, Engelmann MDM, Michaelsen KF, Nielsen MB. Decreased thymus size in formula-fed infants compared with breastfed infants. Acta Paediatr. 1996;85:1029-1032.
5. Goldman AS. Modulation of the gastrointestinal tract of infants by human milk. Interfaces and interactions. An evolutionary perspective. J Nutr. February 2000;130(2S Suppl):426S-431S.
6. Biasucci G, Benenati B, Morelli L, Bessi E, Boehm G. Cesarean delivery may affect the early biodiversity of intestinal bacteria. J Nutr. September 2008;138(9):1796S-1800S.
7. Lebenthal A, Lebenthal E. The ontogeny of the small intestinal epithelium. JPEN J Parenter Enteral Nutr. September-October 1999;23(5 Suppl):S3-S6.
8. Lebenthal E, Lee PC, Heitlinger LA. Impact of development of the gastrointestinal tract on infant feeding. J Pediatr. January 1983;102(1):1-9.
9. Cooperstock MS, Zedd AJ. Intestinal flora of infants. In: Hentges DJ, ed. Human intestinal microflora in health and disease. New York: Academic Press; 1983:79–99.
10. Smith, L. How to Raise A Healthy Child. New York: M Evans and Company; 1996.
11. Dai D, Walker WA. Protective nutrients and bacterial colonization in the immature human gut. Adv Pediatr. 1999;46:353-82.
12. Chana PL, Bik EM, DiGiulio DB, Relman DA, Brown PO. Development of the Human Infant Intestinal Microbiota. PLoS Biology. 1987. Available at: http://www.plosbiology.org/article/info:doi/10.1371/journal.pbio.0050177.
13. D’Adamo P, Whitney C. Live Right for Your Type. New York: G.P. Putnam’s Sons; 2001.
14. Pizzorno J, Murray M. Textbook of Natural Medicine. 3rd ed. Philadelphia: Churchill Livingstone; 2006.
15. Sandberg-Lewis S. Functional Gastroenterology. Portland: NCNM Press; 2009.
16. Shi HN, Walker A. Bacterial colonization and the development of intestinal defences. Can J Gastroenterol. August 2004;18(8):493-500.
17. Gil A, Rueda R. Interaction of early diet and the development of the immune system. Nutr Res Rev. December 2002;15(2):263-292.
18. Jyonouchi H. Non-IgE mediated food allergy. Inflamm Allergy Drug Targets. September 2008;7(3):173-180.[/protected]