Infants, Tweens, and Teens: Do They Hold the Keys to Our Healthy Future?
Jaclyn Chasse, ND
Naturopathic physicians place more emphasis on preventive medicine than many other physician groups. This is extraordinarily important for the prevention of adult-onset chronic diseases such as cancer, diabetes mellitus, cardiovascular disease, and more. With rising rates of chronic disease in the pediatric population, these preventive strategies are increasingly imperative at younger ages. Physicians tend to focus these preventive efforts on what can be done by an individual within a lifetime to affect his or her own health. However, newer research indicates that preventive medicine goes far beyond our own life span and that our current health crises may be tied to the behaviors of our parents and grandparents. This also suggests that to protect the next generation of children (and adults) we must work with our patients in a new way, emphasizing preventive strategies at key time points that have the greatest influence on the next generation.
The paradigm of disease and disease causation has been debated for as long as humans have recognized disease. Recently, the question of nature vs nurture has been at the top of the list for debates. Does genetic predisposition have a greater effect than lifestyle behaviors? How do these factors interact? We now understand that the growing field of epigenetics explains much of the interplay between these 2 factors. The term epigenetics describes heritable changes in gene function that do not involve a change to the actual genome. This is typically accomplished through mechanisms such as modification of DNA function by RNA, acetylation and deacetylation of histones, or methylation and demethylation at a cytosine-guanine dinucleotide of the DNA. These molecular changes do not alter the genome but change the way the genome is read, like a table of contents that directs you to the correct (or incorrect) page of an owner’s manual.
What we have begun to understand is that epigenetic modifications occur with various lifestyle behaviors, dietary choices, and environmental exposures. These epigenetic patterns can be passed from one generation to the next and have a significant effect on health outcomes of children and adults. Most interesting, the greatest periods of influence of these epigenetic modifications occur in childhood.
The 3 key periods for epigenetic imprinting are during the 3 to 4 months before conception, during fetal development, and during puberty. Because the 4 months before conception is the time of final maturation for egg and sperm, parental behaviors during that period may be predictive of their child’s future health. During fetal development, stem cells undergo rapid proliferation and differentiation and are highly susceptible to changes in their environments. In addition, gamete development for girls occurs in utero, so their reproductive future is greatly determined by the environment in the womb. Likewise, puberty is the time of maturation of spermatogenic organs, and exposures during this key time may have a role in testicular health in such a way that gamete development is affected in the long term.
Perinatal Influences
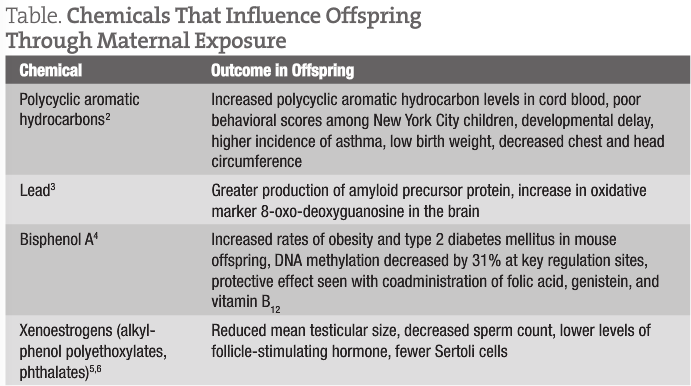
When a woman is pregnant, it makes sense that her behaviors affect the health of the offspring. However, what we are finding out is that it is unlikely that the effects are solely due to direct exposure to the maternal environment. One example of this is the use of diethylstilbestrol during pregnancy. It is well known that offspring born to women who took diethylstilbestrol during pregnancy experienced a higher rate of reproductive tract abnormalities. It has now been observed that the third-generation offspring may also be affected, suggesting an epigenetic influence.1 While diethylstilbestrol is no longer prescribed during pregnancy, exposure to several other chemicals is still prevalent, including bisphenol A, xenoestrogens such as phthalates and alkylphenol polyethoxylates, and environmental contaminants such as lead or polycyclic aromatic hydrocarbons. Maternal exposure to these chemicals has been linked with several effects in the offspring that may influence the first generation and subsequent generations (Table).2-6
Maternal nutrient intake may also dramatically affect the health of the offspring, directly and through epigenetic effects. Maternal undernutrition has been demonstrated to increase the offspring’s risk for obesity, hypertension, mood disorders, impaired glucose and lipid metabolism, and alterations to the hypothalamic-pituitary-adrenal axis and reduced renal function.7,8 Even after 60 years, those conceived to mothers who were undernourished still demonstrated less methylation on marker genes than same-sex siblings who were unexposed.9 While undernutrition, from a caloric perspective, may not seem to be a concern in modern America, we do not yet understand if this process is driven by decreased consumption of calories or by decreased access to key nutrients in the diet. Protein intake has also been extensively investigated for its outcomes on offspring. Studies10-12 have shown that both a high-protein diet (0.45 kg of meat intake daily, with avoidance of carbohydrates) and a low-protein diet (<0.50 g/kg of body weight) modified gene transcription in the offspring, leading to lipid metabolism, vascular dysfunction, increased fat deposition, altered feeding behavior, and increased blood cortisol levels, all of which can affect cardiometabolic risk. The great news is that maternal exposure to specific nutrients, including vitamin A, folic acid, vitamin B12, and genistein, has demonstrated protective effects from many of these perinatal exposures.13
Maternal stress may also have a role in modifying disease risk. Studies14,15 have shown that mothers with high stress levels during pregnancy demonstrated modifications to the infant’s health, including altered immune function, greater risk for atopic disorders, increased salivary cortisol levels, and decreased motor development and learning abilities.
Studies on almost every continent have explored the role that birth weight has on coronary heart disease risk. This correlation is better known as the Barker hypothesis. Data show that people born with a low birth weight are at a significantly higher risk for developing stroke, hypertension, coronary heart disease, and type 2 diabetes mellitus, as well as increased risk for death due to heart disease before age 60 years.16 Low birth weight is determined not by genetic environment but by prenatal environment, including factors such as nutrient availability and oxygen supply; therefore, maternal behaviors have the greatest role in determining birth weight.17 Low birth weight has also been correlated with higher incidences of childhood asthma and acute leukemia in children.17
Prepuberty and Puberty
Behaviors during age 8 to 12 years also demonstrate a significant influence on the health of future offspring, especially for boys. One study18 demonstrated that fathers who started smoking before age 11 years had sons with higher body mass index. In addition, several investigations looking at prepubertal nutrition showed a gender-specific relationship between food supply and cardiovascular health of the offspring, in which protection is conferred to female offspring (but not male offspring) of women with a plentiful food supply during age 8 to 12 years and cardiometabolic protection is conferred to male offspring (but not female offspring) of men with poor food availability during the same age range.18 These transgenerational responses to grandparents’ nutrition prevailed as the main influence on longevity in the second-generation offspring, even more so than their own lifestyle behaviors.18
Naturopathic Conclusion
There is no doubt that behaviors in one’s own life can significantly influence the longevity and health of the individual. However, research demonstrates that additional factors are at play; behaviors during the periods of preconception, pregnancy, and puberty are highly significant. Physicians working with children should be aware of these key influences chiefly because they significantly influence the health of the children in their offices. A firm understanding of what the parental and ancestral health variables are and how those factors affect the children they are treating may give clues to the etiologies of and potential treatments for chronic diseases in their pediatric patients. However, most important is that physicians should take advantage of key periods of epigenetic imprinting to recommend and institute behaviors that will have positive outcomes on future generations, as well as to discourage exposures and behaviors that may cause harm to future generations. This provides an additional, currently overlooked, aspect of preventive medicine that must be addressed if we are to positively affect change in the health status of future generations.
Jaclyn Chasse, ND is a practicing naturopathic physician in New Hampshire. She maintains a busy pediatric practice because of her specialty in infertility. Dr Chasse also works as the manager of educational programs and professional services for Emerson Ecologics (Bedford, New Hampshire), serves as an adjunct faculty member for Bastyr University (Kenmore, Washington), and is active in state and national naturopathic organizations. Dr Chasse enjoys cooking, gardening, reading, and being active with her family.
References
Titus-Ernstoff L, Troisi R, Hatch EE, et al. Birth defects in the sons and daughters of women who were exposed in utero to diethylstilbestrol (DES). Int J Androl. 2010;33(2):377-384.
Rosa MJ, Jung KH, Perzanowski MS, et al. Prenatal exposure to polycyclic aromatic hydrocarbons, environmental tobacco smoke and asthma. Respir Med. 2011;105(6):869-876.
Wu J, Basha MR, Zawia NH. The environment, epigenetics, and amyloidogenesis. J Mol Neurosci. 2008;34(1):1-7.
Dolinoy D, Huang D, Jirtle RL. Maternal nutrient supplementation counteracts bisphenol A–induced DNA hypomethylation in early development. Proc Natl Acad Sci U S A. 2007;104:13056-13061.
Sharpe RM, Fisher JS, Millar MM, Jobling S, Sumpter JP. Gestational and lactational exposure of rats to xenoestrogens results in reduced testicular size and sperm production. Environ Health Perspect. 1995;103(12):1136-1143.
Sweeney T, Nicol L, Roche JF, et al. Maternal exposure to octylphenol suppresses ovine fetal follicle-stimulating hormone secretion, testis size, and Sertoli cell number. Endocrinology. 2000;141(7):2667-2673.
Rosebloom T, deRooij S, Painter R. The Dutch famine and its long-term consequences for adult health. Early Hum Dev. 2006;82:485-491.
St Clair D, Xu M, Wang P, et al. Rates of adult schizophrenia following prenatal exposure to the Chinese famine of 1959-1961. JAMA. 2005;294(5):557-562.
Rosebloom TJ, van der Meulen JH, Osmond C, et al. Coronary heart disease after prenatal exposure to the Dutch famine 1944-45. Heart. 2000;84(6):595-598.
Waterland RA, Garza C. Potential mechanisms of metabolic imprinting that lead to chronic disease. Am J Clin Nutr. 1999;69(2):179-197.
Burdge GC, Delange E, Dubois L, et al. Effect of reduced maternal protein intake in pregnancy in the rat on the fatty acid composition of brain, liver, plasma, heart and lung phospholipids of the offspring after weaning. Br J Nutr. 2003;90(2):345-352.
Herrick K, Phillips DI, Haselden S, et al. Maternal consumption of a high-meat, low-carbohydrate diet in late pregnancy: relation to adult cortisol concentrations in the offspring. J Clin Endocrinol Metab. 2003;88(8):3554-3560.
Dolinoy DC, Huang D, Jirtle RL. Maternal nutrient supplementation counteracts bisphenol A–induced DNA hypomethylation in early development. Proc Natl Acad Sci U S A. 2007;104(32):13056-13061.
Merlot E, Couret D, Otten W. Prenatal stress, fetal imprinting and immunity. Brain Behav Immunol. 2008;22(1):42-51.
Oberlander TF, Weinberg J, Papsforf M, et al. Prenatal exposure to maternal depression, neonatal methylation of human glucocorticoid receptor gene (NR3C1) and infant cortisol stress responses. Epigenetics. 2008;3(2):97-106.
Edwards JW, Stampfer MJ, Manson JE, et al. Birth weight and risk of cardiovascular disease in a cohort of women followed up since 1976. BMJ. 1997;315(7105):396-400.
Kramer MS. Determinants of low birth weight: methodological assessment and meta-analysis. Bull World Health Organ. 1987;65(5):663-737.
Kaati G, Bygren LO, Pembrey M, et al. Transgenerational response to nutrition, early life circumstances and longevity. Eur J Hum Genet. 2007;15(7):784-790.