Melatonin
The Miracle Hormone?
Timothy Schwaiger, ND
For many years the hormone melatonin has been used for a variety of conditions. Although melatonin has primarily been used for the treatment of insomnia, many physicians and researchers have focused on other beneficial therapeutic uses of melatonin.
History of Melatonin
In 1958, a dermatologist by the name of Dr Aaron Lerner discovered melatonin in hopes of being able to treat vitiligo.1 Since that time, melatonin has been studied for various reasons. Originally I used melatonin to help people initiate and maintain sleep. More recently, the hormone has been used as an adjunct to cancer therapy, to improve the immune system, to protect against damage to blood vessels, and to lower blood pressure. In this article I will explore the physiology of melatonin and mechanism by which this hormone can affect so many areas. The US Dietary Supplement Health and Education Act of 1994 approved the use and sale of melatonin because it is found naturally in some foods.2 Prior to that, in 1988, a patent was filed for the use of melatonin as a anti-aging product.3 I am fascinated with not only the many uses of melatonin but also the variety of methods of delivery being explored. Studies explored in this review used oral, transdermal, and intravenous forms of melatonin. Controlled-release and surge-sustained-release forms are also being investigated and utilized by practitioners and researchers.4,5
Melatonin Pathway
Although the majority of melatonin is produced in the pineal gland, it is also synthesized to a lesser degree by other organ systems such as the gastrointestinal system and the heart.6 N-acetyl-5-methoxytryptamine (melatonin) is produced from N-acetylserotonin (NAS). Serotonin is the precursor of NAS and the reaction is catalyzed by the enzyme, serotonin-N-acetyltransferase (SNAT). Melatonin is also converted back to NAS (Figure 1), which is thought to have antidepressant and antihypertensive activity on its own. In addition, NAS may be helpful in treating older individuals for cognitive enhancement.7
Figure 1. The Melatonin Pathway

Oral melatonin has a relatively short half-life, lasting 30 to 60 minutes, and is cleared very efficiently by the liver.8,9 In an attempt to extend the release of melatonin, researchers have developed controlled-release melatonin, as well as transdermal delivery.5,10 One concern about using a sustained-release form of melatonin is the risk of elevating daytime levels of melatonin and potentially interfering with the normal circadian rhythm of the hormone.
Sleep and Dreams
The most important diagnostic consideration in assessing fatigue and insomnia is to evaluate and treat primary sleep disorders. Treating patients with sleep aids without first diagnosing the root cause of the sleep disorder will delay appropriate treatment. Particular patients complaining of daytime fatigue or sleepiness must be evaluated prior to supplemental therapy for enhancing sleep.
Common Sleep Disorders
Insomnia is both a symptom and diagnosis. As a symptom, it is a subjective report of an individual’s inability to fall asleep, stay asleep, or have a high-quality sleep. Insomnia can be transient or become chronic.
Chronic insomnia is highly prevalent, affecting approximately 30% of the general population.11 According to the Sleep Disorders and Research Center in Detroit, MI, criteria for insomnia include (a) difficulty falling asleep, staying asleep, or having non-restorative sleep; (b) the difficulty is present despite opportunities to sleep; (c) the sleep impairment is associated with daytime sleepiness; and (d) the difficulty occurs at least 3 times per week and has been a problem for at least 1 month.9
Transient Insomnia
Changes in one’s environment or stressful events can lead to temporary difficulty in falling asleep. Moving to a new house, a noisy environment, an uncomfortable bed, or too much light in the bedroom can all lead to sleep problems. Once adjustments are made to correct these situations, the insomnia may disappear. On the other hand, if changes are not made, then chronic insomnia may develop.
Primary Sleep Disorders
Primary sleep disorders and circadian rhythm disturbances are common causes of insomnia and must always be ruled out in any case of chronic insomnia. Primary sleep disorders include restless leg syndrome, periodic limb movement disorder (PLMD), sleep-related breathing disorders (snoring, dyspnea, sleep apnea), and narcolepsy.
Restless Leg Syndrome & Periodic Limb Movement Disorder
Restless leg syndrome (RLS) is a common disorder characterized by unusual or unpleasant sensations in the legs (and sometimes in the arms). During sleep, involuntary movements of the limbs may persist, causing the individual difficulty in falling or staying asleep. Most cases of RLS are of unknown cause but can be related to low iron levels, some chronic diseases, or pregnancy.
Sleep Apnea
Sleep apnea can be characterized by the complete cessation of breathing (central sleep apnea) or from an obstruction in the airway (obstructive sleep apnea). Central sleep apnea is less common and occurs primarily in infants and the elderly. Obstructive sleep apnea-hypopnea syndrome (OSAHS) is a common condition and includes the symptoms of snoring, gasping for air during sleep, morning headaches, waking unrefreshed, and excessive daytime sleepiness (EDS).
The conservative treatment for OSAHS is weight loss and sleeping on one’s side, since this condition is usually worse while sleeping supine. The most common medical intervention is nasal continuous positive airway pressure (nasal CPAP), which prevents collapse of the upper airway during sleep.11
Narcolepsy
Narcolepsy is the most common disorder related to disorders of excessive daytime somnolence (EDS) or hypersomnolence. About 60 to 70% of individuals suffering from this disorder will also have cataplexy, which is characterized by sudden muscle weakness, slurred speech, and in some cases total collapse.12 Most of these conditions are treated with medications.
For diagnosis, all of the primary sleep disorders require overnight polysomnography (NPSG). The diagnosis of narcolepsy requires additional daytime testing the day following the NPSG. This test is called the Multiple Sleep Latency Test (MSLT).
Melatonin for Sleep
The first patent for the use of melatonin for inducing sleep was registered in 1995 by Dr Richard Wurtman at the Massachusetts Institute of Technology (MIT).13 Since that time many bottles can be found on the shelves at your favorite market. Doses range from 1 mg to 10 mg and come in tablet, capsule and liquid form.
In a meta-analysis published in 2013,14 melatonin was shown to have positive effects on primary sleep disorders; exogenous administration of melatonin was shown in studies to reduce sleep onset time, increase total sleep time and increase total quality of sleep. In a small study using shift workers that slept during the day, controlled-release transdermal melatonin (2.1 mg) demonstrated improvement of sleep as compared to those taking oral melatonin. Perhaps one of the most interesting studies I came across was one completed in 19 hospitals across England and Wales.15 The researchers demonstrated improvement of total sleep time and reduction of sleep onset time using oral melatonin in children with neurological and developmental disorders.
Patients have reported to me an increased level of dream recall using melatonin in doses of 5 to10 mg. Recently one of my patients reported that she had never recalled her dreams until going on 5 mg of melatonin at bedtime. Of course, not all dreams are pleasant and patients may need to analyze dream content with their ND for reassurance and insight.
Melatonin and Cancer
For some time now, many physicians, including NDs, DOs, and MDs, have been using melatonin as an adjunct in the treatment of solid tumors. Melatonin improves cancer patient outcomes, reduces remission occurrences and offers protection from radiation therapy.16 Typical dose range has been 20 to 40 mg per day.
Melatonin as an Antioxidant
Melatonin, via its catabolites, has been shown to exhibit antioxidant properties.17
N1-acetyl-N2-formyl-5-methoxykynuramine (AFMK) and N1-acetyl-5-methoxykynuramine (AMK), 2 melatonin catabolites, have been described as potent antioxidants. AMK is also a cyclooxygenase (COX) inhibitor. In the conversion of melatonin to cyclic 3-hydroxymelatonin (c3OHM), oxidative processes occur. Other pathways of melatonin directly and indirectly scavenge free radicals such as the hydroxyl radical (•OH) and the superoxide anion (O2• −).
In addition, melatonin has a direct effect on reducing the harmful degradation of lipids (see Figure 2). Increased levels of lipid peroxidation are related to increased oxidative stress. In a study of 20 septic newborns, all newborns receiving 20 mg of melatonin showed reduced levels of lipid peroxidation.19 Increased levels of lipid peroxidation are related to increased oxidative stress.16 In yet another study with neonates who underwent surgery for varoius conditions, intravenous melatonin was found to reduce levels of cytokines and nitric acid synthesis post-surgery.20
Figure 2. Melatonin’s Antioxidant Properties
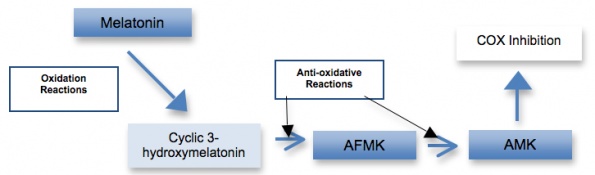
Reactions include both oxidative reactions (eg, lipid peroxidation) and reduction of free radicals. (Adapted from Melatonin: Biological Basis of Its Function in Health and Disease)18
Melatonin and Blood Pressure
Studies are mixed in relation to melatonin and its effect on blood pressure. Grossman and colleagues compared the difference of controlled-release and fast-release melatonin and their effects on systolic and diastolic blood pressures.21 Controlled-release melatonin reduced systolic and diastolic blood pressure by -6.1 mm Hg and -3.1 mm Hg, respectively. Fast-release melatonin reduced systolic and diastolic blood pressures by only -2.1 mm Hg and -1.3 mm Hg, respectively. It should be noted, however, that one of the authors received grant funding from a pharmaceutical company that makes a controlled-release form of melatonin in Europe.
Melatonin has been studied for its ability to counter the side effects of beta-blockers. One major side effect of beta-blockers is reduced sleep quality, as reported by patients using these medications. In a study published in the journal Sleep, those taking 2.5 mg of melatonin for 3 weeks not only reported sleeping better, but night-time polysomnography also demonstrated increased sleep efficiency and increased total sleep time.22

Timothy Schwaiger, ND, graduated with his naturopathic medical degree from Southwest College of Naturopathic Medicine (SCNM) in Tempe Arizona. He holds an undergraduate degree in Behavioral Sciences and a Masters’ degree in Health Services Management and has over 30 years health related experience.
Dr. Schwaiger completed a two-year residency at SCNM in Family Medicine. He served as Associate Professor of Clinical Medicine and Chair of the Department of General Medicine at SCNM. He is Chair of the Laboratory Diagnosis Section for the Naturopathic Physician Licensing Examination (NPLEX), and also serves on its Executive Board.
His practice focuses on integrative family medicine in treating both acute and chronic conditions. He treats individuals with anxiety and depression; sleep disorders, cardiovascular disease, headaches, gastrointestinal disorders, and hormone problems. In addition, he utilizes an integrative approach to cancer treatment. He has had extensive training and clinical experience in Prolotherapy used in the treatment of pain. Dr. Schwaiger also utilizes nutritional IV therapies as part of his treatment approach.
References
- Wurtman RJ. Melatonin as a Hormone in Humans: A History. Yale J Biol Med. 1985;58(6):547-552.
- Dietary Supplement Fact Sheets. Office of Dietary Supplements, National Institutes of Health. http://ods.od.nih.gov. Accessed June 1, 2013.
- Use of melatonin or derivatives thereof for the production of pharmaceutical compositions effective to counteract the effects of aging. Patent File No. WO 1989004659 A1. June 1, 1989. Applicant: Cellena (Cell Engineering) A.G. Inventors: Maestroni G, Pierpaoli W. http://www.google.com/patents/WO1989004659A1?cl=en. Accessed June 1, 2013.
- Gooneratne NS, Edwards AY, Zhou C, et al. Melatonin pharmacokinetics following two different oral surge-sustained release doses in older adults. J Pineal Res. 2012.; 52(4):437–445.
- Grossman E, Laudon M, Yalcin R, et al. Melatonin reduces night blood pressure in patients with nocturnal hypertension. Am J Med. 2006;119(10):898-902.
- Hardeland R. Neurobiology, pathophysiology, and treatment of melatonin deficiency and dysfunction. ScientificWorldJournal. 2012;2012:640389.
- Oxenkrug G, Ratner R. N-acetylserotonin and aging-associated cognitive impairment and depression. Aging Dis. 2012;3(4):330-338.
- Vijayalaxmi, Thomas CR Jr, Reiter RJ, Herman TS. Melatonin: from basic research to cancer treatment clinics. J Clin Oncol. 2002;20(10):2575-2601.
- Srinivasan V, Spence DW, Pandi-Perumal SR, et al. Therapeutic actions of melatonin in cancer: possible mechanisms. Integr Cancer Ther. 2008;7(3):189-203.
- Aeschbach D, Lockyer BJ, Dijk DJ, et al. Use of transdermal melatonin delivery to improve sleep maintenance during daytime. Clin Pharmacol Ther. 2009;86(4):378–382.
- Roth T. Insomnia: definition, prevalence, etiology, and consequences. J Clin Sleep Med. 2007;3(5 Suppl):S7-S10.
- Carney PR. Clinical Sleep Disorders. 2nd ed. Berry RB, Geyer JD, eds. Philadelphia, PA: Lippincott Williams and Wilkins; 2012:185.
- Methods of inducing sleep using melatonin. Patent File No. US5449683. September 12, 1995. Inventor: Wurtman RJ, Original assignee: Massachusetts Institute of Technology. http://www.google.com/patents/US5449683. Accessed June 1, 2013.
- Ferracioli-Oda E, Qawasmi A, Bloch MH. Meta-analysis: melatonin for the treatment of primary sleep disorders. PLoS One. 2013;8(5):e63773.
- Gringras P, Gamble C, Jones AP, et al Melatonin for sleep problems in children with neurodevelopmental disorders: randomized double masked placebo controlled trial. BMJ. 2012;345:e6664. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3489506/pdf/bmj.e6664.pdf. Accessed June 1, 2013.
- Wang YM, Jin BZ, Ai F, et al. The efficacy and safety of melatonin in concurrent chemotherapy or radiotherapy for solid tumors: a meta-analysis of randomized controlled trials. Cancer Chemother Pharmacol. 2012;69(5):1213–1220.
- Russel RJ, Tan DX, Terron MP, et al. Melatonin and its metabolites: new findings regarding their production and their radical scavenging actions. Acta Biochim Pol. 2007;54(1):1-9.
- Pandi-Perumal SR, Cardinali DP, eds. Melatonin: Biological Basis of Its Function in Health and Disease. Austin, TX: Landes Bioscience; 2005.
- Gittto E Karbownik M, Reiter RJ, et al. Effects of melatonin treatment in septic newborns. Pediatr Res. 2001;50(6):756–760.
- Gitto E, Romeo E, Reiter RJ, et al. Melatonin reduces oxidative stress in surgical neonates. JPediatr Surg. 2004;39(2):184–189.
- 21. Grossman E, Laudon M, Zisapel N. Effect of melatonin on nocturnal blood pressure: meta-analysis of randomized controlled trials. Vasc Health Risk Manag. 2011:7:577–584.
- 22. Scheer FA, Morris CJ, Garcia JI, et al. Repeated Melatonin Supplementation Improves Sleep in Hypertensive Patients Treated with Beta-Blockers: A Randomized Controlled Trial. Sleep. 2012;35(10):1395-1402.