Treating Chronic Pain: Reasons to Use a Top-Down System
Naturopathic Perspective
MARK HEISIG, ND
Pain is an interesting phenomenon within the brain and as a part of the human experience. And it’s important to emphasize the human “experience” component, because pain is just that – an experience. This is what makes it perplexing clinically; there is no such thing as a “purely painful” stimulus. What causes me pain in any given scenario may not cause you pain in that same scenario.1
For example, think about the soldier who’s been hit by explosively hot shrapnel, yet continues to run, move, and perform his duties without blinking or hesitation. Now think about yourself walking to get a late-night snack, stubbing your pinky toe on the coffee table, and subsequently crying out to the heavens and cursing your table to Hell. This is a hilariously embarrassing comparison, but a beautiful example of the context-dependency in the experience of pain.
Understanding this context (and some basic neuroanatomy) can be useful in leveraging some very simple clinical strategies to make marked improvements in treatment outcomes.
Biomechanics or the Egg?
Biomechanics or the egg (the “egg” being “drugs/surgery”) is the age-old question. In America, we spend up to $635 billion per year on over 100 million patients suffering from chronic pain.2 Moreover, “Pharmacologic and interventional treatments for chronic pain often provide no reduction or only a small reduction in pain and are often judged by the patient to be inadequate.”3
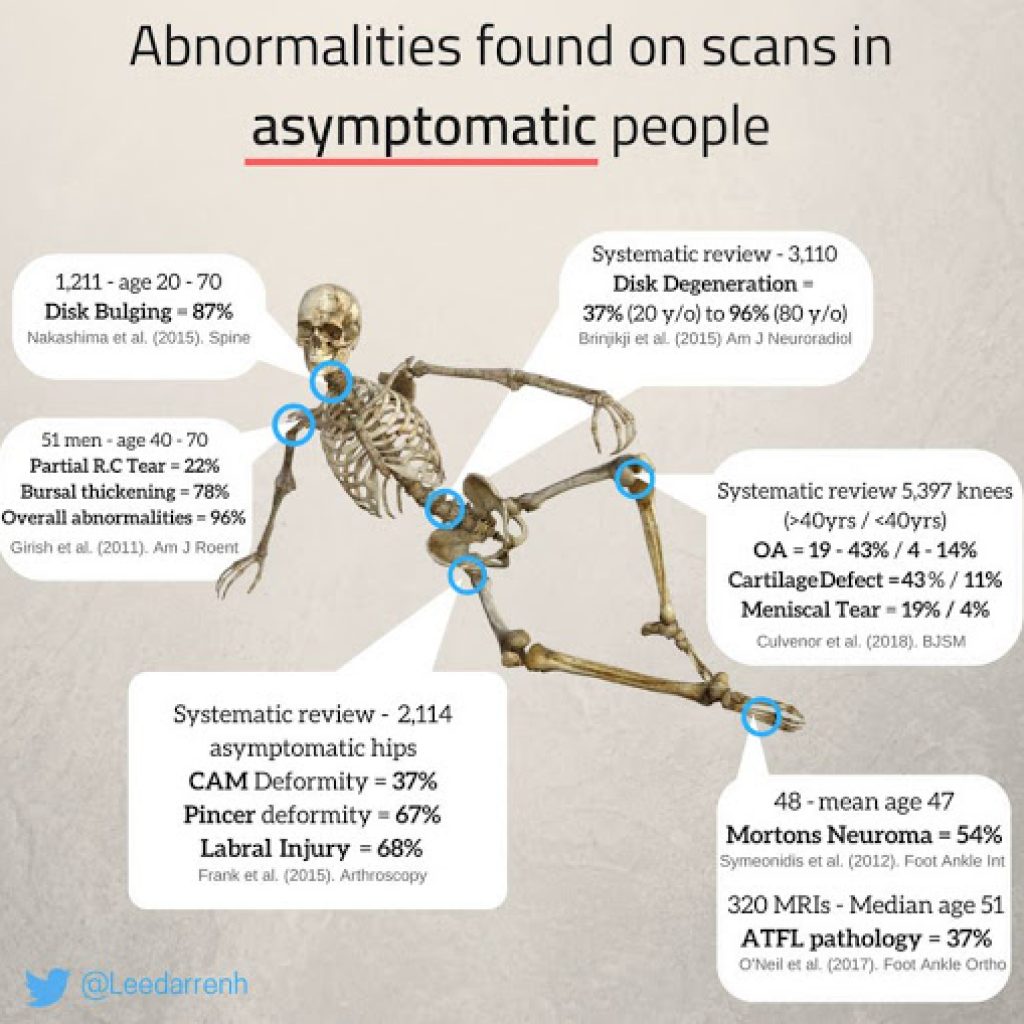
We have a conventional system that reflexes to pharmaceuticals for patients (eg, NSAIDs, antidepressants, anti-epileptics, opioids, etc), guided by “Ladder” and “Trolley” models of analgesic prescription.4,5 On the flip side of this “pain coin,” many practitioners (DC, DO, DPT, ND, AT, etc) will evaluate and assess for biomechanical dysfunction as the root of chronic pain. The problem is that many folks walk around both asymptomatic and completely biomechanically dysfunctional (see Figure 1).
Figure 1. Asymptomatic Biomechanical Dysfunction
So, which is “right?” Ask each of the $635 billion spent, and they’ll say neither. Pain is requiring us to adopt a new lens in order to achieve better outcomes, to move from “management” to “treatment.”
Defining the Parameters
One of the big motivators of seeking pain treatment is the pain beginning to interfere with movement – the ability to walk, play, drive, sit, stand, sleep, etc. Pain interferes with movement. Therefore, pain management should be gauged by the amount of pain-free range of motion gained by any given therapy.
If the treatment doesn’t help you move pain-free, did it in fact treat your pain? That is the ultimate parameter in these chronic, non-cancer, musculoskeletal pain conditions.
Pain treatment is often focused on “finding the pain generator,” ie, finding the tissue in question that is the root of all evil in the patient’s pain condition.
The idea that pain is only perceived when it is being generated (ie, nociception) is “bottom-up” thinking, a nociception-driven thought process. However, if you turn the problem on its head, we have to look at anti-nociception. We need to ask, “How are the anti-nociceptive mechanisms working in this patient?”
Pain is perceived in the cortex by passing from a nociceptor to the spinal cord, up the cord, through the thalamus, shot to different regions of the brain stem and cortex, and then finally perceived as “ouch!” by our conscious minds. We treat this system by2,3:
- NSAIDs, acetaminophen
- local injections
- manipulations, soft-tissue release
- etc… The treatment is peripheral (bottom-up)
Of note, we almost exclusively use this bottom-up system to evaluate and examine a patient. We perform a physical exam and orthopedic maneuvers to recreate the pain and thus find the pain generator. We treat the site (or close to it, eg, fascial connections) of pain.
What is not often talked about is the descending monoaminergic (anti-nociceptive) system.6,7 This is a system that shuts down the pain when it is no longer useful or when it is inhibiting our ability to survive when running from the lion. We treat this system with top-down, central treatments2,3:
- opioids
- tricyclic antidepressants
- cognitive behavioral therapy (CBT) and mind-body interventions
- etc…
This top-down pain modulation system includes our periaqueductal gray (PAG), locus ceruleus (LC), and nucleus raphe magnus (NRM). These nuclei work together to regulate the pain signal(s) coming up from the nociceptors and to modulate the sympathetic response as a part of the pain response.
Of note, we almost never evaluate or examine the integrity of this top-down system.
Pain & Neuroanatomy Basics
Concept 1: Threat & the Neuromatrix
It’s really important to drive home the concept that pain is an experience generated in the brain that is based upon sensory input to the cortex combined with context and interpretation of that sensory information. The unique soup of stimulus and circumstance that elicits the experience of pain constitutes a person’s unique neurosignature within their neuromatrix.8
Much of the affective context that is evaluated for in a patient in pain is “threat.” Perceived by the anterior cingulate cortex (ACC) and salience networks, your brain is constantly looking for “error” that can disrupt your ability to move safely through the world.9 Remember, keep it simple. Clinically, “threat” is anything that impairs one’s ability to exist and live safely in the world.
In the context of chronic pain states, “threat” is central or peripheral sensitization, aberrant proprioception, altered vestibular integration, anxiety/depression, etc. “Threat” lowers thresholds for the experience of pain.
Concept 2: Sensation, Integration, Output
“Sensation, integration, and output” is the name of the neurology game. When nociception, especially chronic, is introduced to the sensation component of the equation, integration and output are altered. This then influences feedback, which further alters integration and output.10 (Now we’ve got a creaky, painful knee and walk with a limp.)
Specifically, nociception alters sensation, integration, and motor output while also activating the sympathetic nervous system, which makes sensitization of the painful site more likely. The chronic nociception and altered sensation eventually distort the cortical representation (somatotopic map) of the body region,11,12 which further impairs descending pain modulation and blood flow regulation (eg, reflex sympathetic dystrophy/complex regional pain syndrome). Think about it this way: “You can’t inhibit pain in your hand if you don’t accurately know where it is in space.”
Clinical question: Can you really improve proprioception, then, in a painful body region if the neuroanatomy of pain distorts proprioception to begin with? Can you build a new and improved somatotopic map by using a distorted somatotopic map? Put simply, can you use a broken map to build a good map?
Concept 3: Oculomotor Function as a Window into Brain Function
More than 50% of the known pathways in the brain are devoted to vision and eye movements. The eyes and oculomotor function are increasingly becoming a validated clinical window into brain function.13,14 The idea is that if we can detect an aberrancy “outside” (eye movements), we can trace that back to an aberrancy “inside” (brain). In pain states, we see altered coupled head-eye movement, smooth pursuits, and saccades.14-17 This makes oculomotor examination a viable, bedside means of assessing cortical function and its relation to pain states. However, that is beyond the scope of this article.
“Top-down” Evaluation & Management
For a complete evaluation of pain, we should not only investigate for “pain generators” peripherally; we should also:
- Evaluate for anti-nociceptive integrity, centrally
- Evaluate for accurate body schema
- Find ways to reduce “threat” in the system
Evaluating for anti-nociceptive integrity
Detailing this approach is beyond the scope of this article. In short, it includes a detailed examination of the frontal lobe, cerebellum, midbrain, pons, and medulla through oculomotor function, posture, and gait.14,18
Evaluating for accurate body schema
The body is loaded with maps. You have your somatotopic map in the parietal lobe, a visual map in the superior colliculus, an auditory map in the inferior colliculus, 3 maps of your body in the cerebellum, and so on. Knowing where you are in space is critical to survival as well as and pain-free movement.19,20 Here are just some ways to evaluate body schema/proprioception is a relevant problem in your patient:
- Cerebellar testing
- Romberg test
- Touch/Point localization
- Joint position error testing (JPET)21-23
- Fukuda step test
When you find an aberrant map, you will know that part of that patient’s therapy now has to include “re-building” the map. The alternative is to risk staying stuck on a treadmill of altered sensation, integration, and output. One of the ways we avoid that treadmill and improve therapy is by building upon pain-free range of motion in non-threatening ways.
Finding ways to reduce threat
Reducing threat in your patient may be a mind-body or psychology referral to work on anxiety, depression, trauma, etc, while you continue to work on metabolic or physical rehab.
Within the physical realm, from a non-energetic and a neuroanatomically-driven perspective, we need to encourage proprioceptively accurate, safe, pain-free movement. This can include:
- Guided imagery/Left-Right discrimination24
- Mirror therapy25
- Isometric contraction/coordination26
- Laser-guided visual feedback27
The idea is to take the “good, pain-free map” and grow it – to make the demarcations and cortical body representations sharper and more accurate.
Putting It All Together
Pain is a unique, individual experience that is context and “threat-dependent.” Part of the problem in chronic pain is a distorted body schema, sensitization, and chronic sympathetic activation. One way to break this cycle is to stop thinking about the site of pain and to start thinking about the perception and modulation of pain. A big piece of that is reducing threatening movement and improving body schema to increase pain-free range of motion.
For example, a patient with persistent knee pain years after an ACL injury likely has altered sensory-motor integration and a distorted perception of their knee in space. Testing may reveal poor stability with eyes closed during the Romberg test, pronation and an adductory twist with gait, and impaired touch localization at the knee joint. This person does not know where their knee is in space. Therapy may include:
- Elastic taping strategies for passive sensory feedback from the knee28,29
- Isometric contraction in a pain-free degree of flexion, gradually expanding towards greater pain-free flexion (or extension)
- Laser-guided visual feedback during traditional knee physical rehab exercises
- Etc…
The application is simple. The lens is different.
A special thanks to The Carrick Institute for outlining this beautiful paradigm of pain evaluation and management.
Reference:
- Kandel ER, Schwartz JH, Jessel TM, et al, eds. Principles of Neural Science. 5th ed. New York, NY: McGraw-Hill; 2013.
- Bonakdar RA. Integrative Pain Management. Med Clin North Am. 2017;101(5):987-1004.
- Finnerup NB. Nonnarcotic Methods of Pain Management. N Engl J Med. 2019;380(25):2440-2448.
- Cuomo A, Bimonte S, Forte CA, et al. Multimodal approaches and tailored therapies for pain management: the trolley analgesic model. J Pain Res. 2019;12:711-714.
- Yang J, Bauer BA, Wahner-Roedler DL, et al. The Modified WHO Analgesic Ladder: Is It Appropriate for Chronic Non-Cancer Pain? J Pain Res. 2020;13:411-417.
- Benarroch EE. Descending monoaminergic pain modulation: bidirectional control and clinical relevance. Neurology. 2008;71(3):217-221.
- Bannister K. Descending pain modulation: influence and impact. Curr Opin Physiol. 2019;11:62-66.
- Melzack R. Pain and the neuromatrix in the brain. J Dent Educ. 2001;65(12):1378-1382.
- Fuchs PN, Peng YB, Boyette-Davis JA, Uhelski ML. The anterior cingulate cortex and pain processing. Front Integr Neurosci. 2014;8:35.
- Nijs J, Daenen L, Cras P, et al. Nociception affects motor output: a review on sensory-motor interaction with focus on clinical implications. Clin J Pain. 2012;28(2):175-181.
- Haggard P, Iannetti GD, Longo MR. Spatial sensory organization and body representation in pain perception. Curr Biol. 2013;23(4):R164-R176.
- Schabrun SM, Hodges PW, Vicenzino B, et al. Novel adaptations in motor cortical maps: the relationship to persistent elbow pain. Med Sci Sports Exerc. 2014;5(1):1-34.
- Felleman DJ, Van Essen DC. Distributed hierarchical processing in the primate cerebral cortex. Cereb Cortex. 1991;1(1):1-47.
- Shaikh AG, Zee DS. Eye Movement Research in the Twenty-First Century – a Window to the Brain, Mind, and More. Cerebellum. 2018;17(3):252-258.
- Johnston JL, Daye PM, Thomson GT. Inaccurate Saccades and Enhanced Vestibulo-Ocular Reflex Suppression during Combined Eye–Head Movements in Patients with Chronic Neck Pain: Possible Implications for Cervical Vertigo. Front Neurol. 2017;8:23.
- Mosimann UP, Müri RM, Felblinger J, Radanov BP. Saccadic eye movement disturbances in whiplash patients with persistent complaints. Brain. 2000;123(4):828-835.
- Ruscheweyh R, Fritz A, Eggert T, et al. Oculomotor Disturbances in Patients with Chronic Nonspecific Spinal Pain. Pain Medicine. 2018;19(10):2031-2038.
- Takakusaki K. Functional Neuroanatomy for Posture and Gait Control. J Mov Disord. 2017;10(1):1-17.
- Blakeslee S, Blakeslee M. The Body Has a Mind of Its Own: How Body Maps in Your Brain Help You Do (Almost) Everything Better. New York, NY: Random House, Inc; 2007.
- Moseley GL. Graded motor imagery is effective for long-standing complex regional pain syndrome: a randomised controlled trial. Pain. 2004;108(1-2):192-198.
- de Vries J, Ischebeck BK, Voogt LP, et al. Joint position sense error in people with neck pain: a systematic review. Man Ther. 2015;20(6):736-744.
- Ramos MM, Carnaz L, Mattiello SM, et al. Shoulder and elbow joint position sense assessment using a mobile app in subjects with and without shoulder pain-between-days reliability. Phys Ther Sport. 2019;37:157-163.
- Quartey J, Ernst M, Bello A, et al. Comparative joint position error in patients with non-specific neck disorders and asymptomatic age-matched individuals. S Afr J Physiother. 2019;75(1):568.
- Herrador Colmenero L, Perez Marmol JM, Martí-García C, et al. Effectiveness of mirror therapy, motor imagery, and virtual feedback on phantom limb pain following amputation: A systematic review. Prosthet Orthot Int. 2018;42(3):288-298.
- Abolfazli M, Lajevardi L, Mirzaei L, et al. The effect of early intervention of mirror visual feedback on pain, disability and motor function following hand reconstructive surgery: a randomized clinical trial. Clin Rehabil. 2019;33(3):494-503.
- Rio E, Kidgell D, Purdam C, et al. Isometric exercise induces analgesia and reduces inhibition in patellar tendinopathy. Br J Sports Med. 2015;49(19):1277-1283.
- Lam AW, HajYasien A, Kulic D. Improving rehabilitation exercise performance through visual guidance. Conf Proc IEEE Eng Med Biol Soc. 2014;2014:1735-1738.
- Bischoff L, Babisch C, Babisch J, et al. Effects on proprioception by Kinesio taping of the knee after anterior cruciate ligament rupture. Eur J Orthop Surg Traumatol. 2018;28(6):1157-1164.
- Liu K, Qian J, Gao Q, Ruan B. Effects of Kinesio taping of the knee on proprioception, balance, and functional performance in patients with anterior cruciate ligament rupture: A retrospective case series. Medicine (Baltimore). 2019;98(48):e17956.

Mark Heisig, ND, is a naturopathic physician based in Scottsdale, AZ. He is a graduate of Bastyr University in Seattle, WA. Dr Heisig specializes in integrative neurology and metabolic medicine, striving to be a leader in integrative concussion recovery and performance. He inspires athletes and motivated individuals with simplicity and innovation to achieve new levels of higher quality performance in sport and life.