Kulveen Virdee, ND
Primum Non Nocere
The long-term use of immunosuppressive medications puts transplant recipients at risk of BK nephropathy, a viral complication of organ transplant that can lead to renal dysfunction or failure. Current management strategies include early detection through monitoring of plasma and urine levels of BK virus and reduction of immunosuppressive medications. Reducing immunosuppressive medications increases the risk of acute allograft rejection. The case is presented of a 20-year old male, born with posterior urethral valves. He had a prior history of acute allograft failure and developed BK nephritis. Although the standard recommendation is to reduce immunosuppressive medication dosing until the viral load clears, due to his history of allograft rejection, he chose to combine natural therapies to enhance his allograft survival. After 7 weeks of combination treatment with naturopathic interventions that included immunomodulating herbs, renal support, and antiviral therapies, as well as pharmacological interventions, he experienced a reduction of his BK nephritis and improvement in renal function.
Background
BK virus is a member of the polyoma family of viruses. Primary infections with BK virus are generally asymptomatic and occur in childhood. If symptomatic, infected individuals will experience fever and an upper respiratory tract infection. After infection, the virus remains latent in organ tissues including the kidney, lung, eye, liver and brain. BK virus may reactivate and cause symptoms in immunocompromised patients including HIV-infected patients and transplant recipients.1 Rates of infection with BK virus range from 1-10% of transplant recipients,2 and the prevalence of BK virus 1 year after transplantation is about 20%.3,4
BK virus infection causes inflammation of tubular cells, necrosis, and deterioration of allograft function in as many as 80% of cases .2,5-10 In 1 study, infection with BK virus was associated with a 40% incidence of graft loss.6 Renal transplant recipients who have a history of transplant rejection or who are being managed with tacrolimus or mycophenolic acid are at an increased risk of developing BK viral disease.2,11 BK nephritis commonly presents as asymptomatic with a gradual rise in creatinine that mimics acute allograft rejection. Clinicians must differentiate between viral-induced kidney damage and acute rejection since the former requires reduction of immunosuppressive medications while the latter requires stronger dosing of the immunosuppressive medications.5 When an individual is prone to allograft rejection, reduction of immunosuppressive medications can put the allograft at risk of rejection;2,11 nevertheless, there is limited research on other treatments for BK nephropathy.5 The success rates of protocols for BK virus are variable and estimates of graft loss risk range from <10% to >80%.11,12 Treatment continues until the viral load, assessed through BK virus polymerase chain reaction (BKV PCR), is either undetectable or falls below 10,000 viral copies/mL, the threshold associated with nephropathy. The incidence of BK nephropathy has declined since the early detection and intervention protocols have been set forth, however much study is still required.9
Case Presentation
The patient was a 20-year-old Caucasian male with a history of end-stage renal disease as a result of congenital posterior urethral valve defect. His first transplant was at the age of 8 and was donated by his father. This kidney functioned adequately for 9 years prior to rejection. His second transplant was from an unrelated donor and was complicated by fevers and the development of an acute Epstein barr virus (EBV) infection. This was successfully treated with plasmapheresis and intravenous immunoglobulin. After the second transplant, he experienced multiple episodes of acute rejection, which were managed through combination immunosuppressive therapy with tracolimus, prednisone, and modafinil. In December of 2014, his biopsy indicated borderline rejection, for which he was treated with a 6-week prednisone taper and an increase in maintenance prednisone from 5 mg to 10 mg QD. From that time onward, he experienced fluctuations in both EBV and BK virus serum viral loads. In May of 2015, the diagnosis of BK nephropathy was made after noting increases in his creatinine levels. He was referred for a renal biopsy, which showed interstitial fibrosis in about 60% of the sample. His nephrologist discontinued mycophenolate, reduced the dosage of tacrolimus by 50%, and initiated weekly monitoring of tacrolimus levels. Three days after diagnosis, the patient initiated treatment with me.
Treatment
Prior to beginning treatment, naturopathic interventions needed to be assessed for potential interactions with the patient’s medication. Theoretical concerns regarding immune over-stimulation, due to his history of allograft rejection and his reduced dosages of immunosuppressive medications, also needed to be considered. After careful consideration and consultation with his nephrologist, the following therapies were selected.
Immunomodulating Herbs
Immunomodulating herbs are known to regulate immune activity by reducing hypersensitivity reactions while strengthening resistance to infections. A growing body of evidence supports a role for immunomodulating herbs in maintaining allograft survival in transplant patients. Cordyceps sinensis is widely used in China as an adjuvant to immunosuppressive treatment in kidney transplant recipients.13 It is thought that C. sinensis exerts its beneficial effects through its antioxidant, anti-inflammatory, and immune regulating properties, preventing renal interstitial fibrosis and renal tubal impairment.15 In renal failure patients, it has been shown to increase creatinine clearance, reduce serum creatinine, and reduce proteinuria.14 Preliminary clinical research suggests that it may reduce renal toxicity of calcineurin inhibitors (CNI) including tacrolimus, and can boost allograft survival while reducing the need for CNI.13,15 Based on this evidence, C. sinensis was selected to improve allograft survival, reduce the need for immunosuppressive medications, improve kidney function in spite of extensive scarring caused by BKV, and stimulate antiviral immune activity.
Curcuma longa has demonstrated immunosuppressive potential against overexpressed TH1 cytokines, which are often elevated in renal transplant patients and contribute to allograft rejection. It has been found to be safe to use along with cyclosporine and cyclosporine analogs. Finally, C. longa reduces inflammation associated with acute and chronic allograft rejection.16,17 Chronic allograft rejection is also associated with reduced blood flow to the kidneys, resulting in inflammation and oxidative damage. Curcumin has been found to help prevent tissue injury through its antioxidant activity.18
Renal Support
Due to the extensive scarring caused by the initial allograft rejection episodes and the BK viral nephropathy in his only kidney, the goal of nutritional support was to enhance his kidney function.
Silybum marianum’s active constituent, silymarin, has a small body of evidence demonstrating a nephroprotective property in diabetic nephropathy and in patients using nephrotoxic medications, including CNIs.19,20
L-Carnitine has been studied for its ability to improve kidney function and reduce side effects in hemodialysis patients.21 One proposed mechanism of action is through improving the transport of long-chain fatty acids into the mitochondria for energy production.22
Omega-3 fatty acids have been found to reduce systemic inflammation and proteinuria in renal transplant patients.23 The omega-3 fatty acids serve as precursors to anti-inflammatory prostaglandins. Persistent systemic inflammation is common in transplant recipients and is thought to contribute to allograft dysfunction.
Coenzyme Q10 is well known for its antioxidant properties. Increased oxidative stress is noted in renal transplant patients. One study noted that coenzyme Q10 improved multiple markers of antioxidant status and lipid peroxidation in renal transplant patients.24
Anti-viral therapy
Many anti-viral herbs help to overcome viral infections by stimulating natural killer and/or T cell activity. These cells are targets of immunosuppressive therapies due to their involvement in allograft rejection, and a goal of not stimulating these immune cells guided the selection of an anti-viral agent.
Colloidal silver was selected for its known activity against viral particles. Human research supporting the use of colloidal silver for infections is limited; nevertheless, silver nanoparticles are known to be antimicrobial and are widely used to prevent infections in burn wounds and to sterilize surgical and dental equipment. Growing research indicates that silver nanoparticles may be effective in reducing infectivity of HIV.25
Outcomes
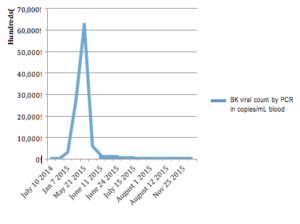
Over a period of 3 months, our patient demonstrated incremental reductions in BK viral count. After 7 weeks of combination therapy, his levels fell below the threshold of 10,000 copies/mL, indicating that BK nephropathy had resolved. After 11 weeks of treatment, his level fell to approximately 1,000 copies/mL.
The patient began experiencing pruritus 1 week after initiating treatment. After ruling out other causes, including uremia, we began assessing whether any supplements could have been contributing to the itching. We suspected the colloidal silver was causing itching and his dose was reduced from daily to 2-3 times per week. Despite this dose reduction, viral titers continued to decline. In early August 2015, the patient discontinued colloidal silver and viral titers increased by 1,851.
As of July 2016, he continues the recommended therapies, with the exception of colloidal silver, and has not had any episodes of acute allograft rejection or recurrence of BK nephropathy. He continues on the lower immunosuppressive dose, and his serum creatinine has decreased to 2.5.
Discussion
This case demonstrates a successful reduction of BK viral titers and resolution of BK nephropathy through an integrative treatment approach. Because clear data regarding the clearance rate of BK virus with reduction of immunosuppressant medications is lacking, it is difficult to compare the response in this case to a standard; however, the nephrologist involved in the case remarked anecdotally that, typically, a positive response to immunosuppressive drug dose reduction is seen after a few months to almost a year. There are retrospective mathematic models demonstrating that it may take anywhere from 7 to 20 weeks for BK viremia to clear.26 Limiting the time between identification of BK nephropathy and reduction of BK virus numbers is critical in prevention of irreversible kidney damage. In this case, adjunctive therapy with coenzyme Q10, curcumin, and fish oil may have been helpful in lowering viral load and reducing kidney inflammation and damage. This may have been especially important given the history of acute allograft rejection prior to the development of BK nephropathy. Finally, our patient has been able to maintain healthy allograft function with continuation of the immunomodulating and nephroprotective support while on a reduced dose of immunosuppressive medications. This is important since many transplant patients develop nephrotoxicity within 10 years of beginning CNI therapy.12 This case report provides evidence that integrative approaches to post-transplant management may contribute to reducing the risk of opportunistic infection with BK virus, improving allograft function, and preventing allograft rejection.
References:
- Reploeg MD, Storch GA, Clifford DB. BK virus: A clinical review. Clin Infect Dis. 2001;33(2):191–202.
- Hirsch HH, Drachenberg C, Steiger J, Ramos E. Polyomavirus-associated nephropathy in renal transplantation: critical issues of screening and management. Adv Exp Med Biol. 2006;577:160–173.
- Hussain S, Bresnahan B, Cohen EP, Hariharan S. Rapid kidney allograft failure in patients with polyomavirus nephritis with prior treatment with antilymphocyte agents. Clin Transplant. 2002;16(1):43–47.
- Brennan DC, Agha I, Bohl DL, et al. Incidence of BK with tacrolimus versus cyclosporine and impact of preemptive immunosuppression reduction. Am J Transplant. 2005;5(3):582–594.
- Bohl DL, Brennan DC. BK virus nephropathy and kidney transplantation. Clin J Am Soc Nephrol 2007;2(Supplement 1):S36–S46.
- Nickeleit V, Hirsch HH, Binet IF, et al. Polyomavirus infection of renal allograft recipients from latent infection to manifest disease. J Am Soc Nephrol. 1999;10(5):1080–1089.
- Binet I, Nickeleit V., Hirsch HH, et al. Polyomavirus disease under new immunosuppressive drugs: a cause of renal graft dysfunction and graft loss. 1999;67(6):918–922.
- Hirsch HH, Steiger J. Polyomavirus BK. Lancet infect Dis. 2003;3(10):611–623.
- Randhawa PS, Finkelstein S, Scantlebury V, et al. Human Polyoma Virus-Associated Interstitial Nephritis in the Allograft Kidney1. 1999;67(1):103–109.
- Ramos E, Drachnberg CB, Papadimitriou JC, et al. Clinical course of polyoma virus nephropathy in 67 renal transplant patients. J Am Soc Nephrol. 2002;13(8):2145–2151.
- Garces JC. BK virus-associated nephropathy in kidney transplant recipients. Ochsner J. 2010;10(4):245–249.
- Vasudev B, Hariharan S, Hussain SA, et al. BK virus nephritis: Risk factors, timing, and outcome in renal transplant recipients. Kidney Int. 2005;68(4):1834–1839.
- Hong T, Zhang M, Fan J. Cordyceps sinensis (a traditional Chinese medicine) for kidney transplant recipients. The Cochrane Database Syst Rev. 2015;10:CD009698.
- Zhang,HW, Lin ZX, Tung YS, et al. Cordyceps sinensis (a traditional Chinese medicine) for treating chronic kidney disease. The Cochrane Database of Syst Rev. 2014;12:CD008353.
- X, Li L. Cordyceps sinensis in protection of the kidney from cyclosporine A nephrotoxicity. Zhonghua yi xue za zhi. 1993;73(7):410-412.
- Bharti AC, Panigrahi A, Sharma PK, et al. Clinical relevance of curcumin-induced immunosuppression in living-related donor renal transplant: an in vitro analysis. Exp Clin Transplant. 2010;8(2):161–171.
- Shoskes D, Chantale L, Cruz-Corerra M, et al. Beneficial effects of the bioflavonoids curcumin and quercetin on early function in cadaveric renal transplantation: A randomized placebo controlled trial. 2005;80(11):1556–1559.
- Aggarwal BB, Harikumar KB. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int J Biochem Cell Biol. 2009;41(1):40–59.
- Rafieian-Kopaie M, Nasri H. Silymarin and diabetic nephropathy. J Renal Inj Prev 2012;1(1):3–5.
- Yarnell EL. Botanical medicines used for kidney disease in the United States. Iran J Kidney Dis. 2012;6(6):407–418.
- Kendler BS. Carnitine: An overview of its role in preventive medicine. Prev Med.1986;15(4):373–390.
- Matera M, Bellinghieri G, Constantino G, et al. History of L-carnitine: implications for renal disease. J Renal Nutr. 2003;13(1):2–14.
- Sabbatini M, Apicella L, Cataldi M, et al. Effects of a diet rich in N-3 polyunsaturated fatty acids on systemic inflammation in renal transplant recipients. J Am Coll Nutr. 2013;32(6):375–383.
- Długosz A, Kuzniar J, Sawicka E, et al. Oxidative stress and coenzyme Q10 supplementation in renal transplant recipients. Int Urol Nephrol. 2004;36(2):253–258.
- Lara HH, Ayala-Nuñez NV, Ixtepan-Turrent L, Rodriguez-Padilla C. Mode of antiviral action of silver nanoparticles against HIV-1. J Nanobiotechnology. 2010;8:1.
- Funk GA, Steiger J, Hirsch HH. Rapid dynamics of polyomavirus type BK in renal transplant recipients. J Infec Dis. 2006;193(1):80–87.
 Kulveen Virdee, ND, is a licensed naturopathic physician in the state of Connecticut. She earned her doctorate from Southwest College of Naturopathic Medicine and completed her residency at Southwest Naturopathic Medical Center. She is the current professor of medical immunology at the University of Bridgeport College of Naturopathic Medicine. In addition, Dr Virdee is passionate about teaching her local community about natural medicine through her public radio show entitled Seeking Wellness: Our Bodies Explained. Her hobbies include exploring the great outdoors and spending time with her cats.
Kulveen Virdee, ND, is a licensed naturopathic physician in the state of Connecticut. She earned her doctorate from Southwest College of Naturopathic Medicine and completed her residency at Southwest Naturopathic Medical Center. She is the current professor of medical immunology at the University of Bridgeport College of Naturopathic Medicine. In addition, Dr Virdee is passionate about teaching her local community about natural medicine through her public radio show entitled Seeking Wellness: Our Bodies Explained. Her hobbies include exploring the great outdoors and spending time with her cats.