Ronald Steriti, ND, PhD; Jonathan V. Wright, MD
The enzyme aromatase converts testosterone into estradiol and converts androstenedione into estrone. The mechanism of aromatase metabolism is shown in the Figure.
Treatment for Increased Estrogen and Decreased Testosterone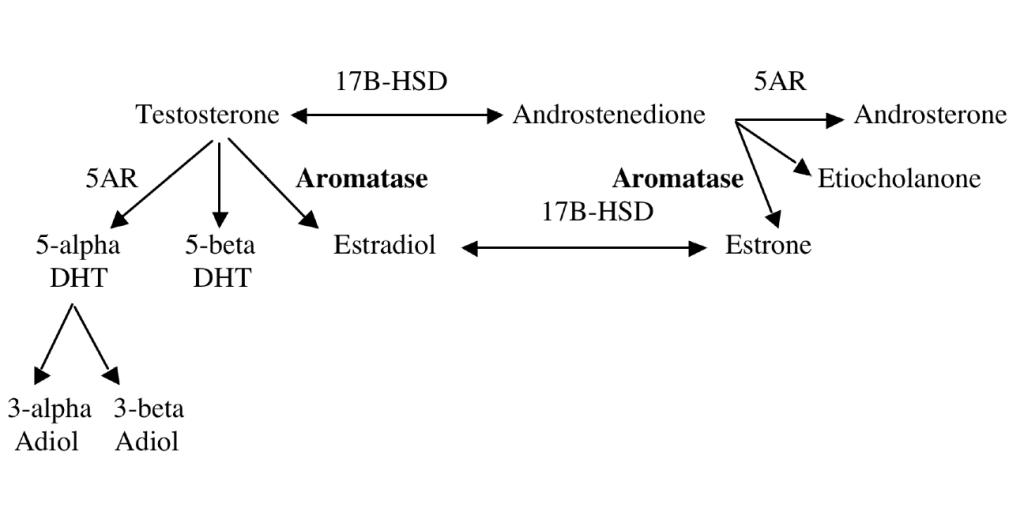
Inappropriate diet is considered the main cause of insulin resistance, which is the primary pathophysiologic feature of metabolic syndrome. Increased insulin levels stimulate aromatase activity.1,2
In obese men, increased aromatase activity results in reduced testosterone and increased estradiol and estrone concentrations, ultimately resulting in a decrease in muscle mass and an increase in total fat mass. Increased total fat mass contributes to oxidative stress, inflammation, and further increased aromatase activity (wrong diet à hyperinsulinism à hyperaromatization à excess obesity).3-5
Cohen3 proposed that aromatase inhibitors could be potential therapeutic agents for men with obesity. Leder6 recommended their use during testosterone therapy in older men to prevent adverse effects of estradiol.
Male gynecomastia is usually caused by increased estrogen activity, decreased testosterone activity, or the use of numerous medications. Excess aromatase activity is also a cause of gynecomastia.7-12
Hyperaromatization
Hyperaromatization is a term used to describe excessive aromatase activity, which results in conversion of testosterone into estrogen. Although possible in both sexes, hyperaromatization is particularly found in men and often accompanies low testosterone concentration and obesity related to metabolic syndrome and insulin resistance.13
Myomin and Chrysin
Available natural formulas that help reverse hyperaromatization include myomin and chrysin. Myomin is a combination of Smilax glabra Roxb, Curcuma zedoria, Cyperus rotundus, and Aralia dasyphylla Mig.14
A fluorescent antibody study15 in animals showed that myomin significantly reduced the population of hepatic aromatase within 30 days. In another study,16 myomin significantly inhibited aromatase expression in in vivo ovary, ectopic endometrium, and in vitro–cultured primary ovarian granulosa cells of rats.
Chrysin is a naturally occurring flavone. It is chemically extracted from the blue passion flower (Passiflora caerulea).17-20
While these aromatase inhibitors of natural origin appear to be harmless, they are not known to correct hyperinsulinism. It is important for men with excess testosterone-to-estrogen transformation to undergo testing for insulin resistance21 and, if found, to follow appropriate diet, exercise, and supplementation to decrease or stop hyperinsulinism, thus reducing or eliminating the excess conversion and in many cases rendering aromatase inhibitor use of any sort less necessary or unnecessary at all.
Turnera diffusa
A study22 showed that Turnera diffusa (damiana) extract and 2 isolated compounds (pinocembrin and acacetin) could significantly suppress aromatase activity. Apigenin 7-glucoside, Z-echinacin, and pinocembrin demonstrated estrogenic activity.
Aromataste Inhibitor Drugs
Aromatase inhibitor drugs (patent pharmaceuticals) are “approved” for estrogen receptor positive (ER+) early breast cancer and following Tamoxifen therapy. They include Aromasin ® (exemestane), an irreversible steroidal inhibitor; Femara ® (letrozole) and Arimidex ® (anastrozole), which are non-steroidal inhibitors. Aromatase inhibitors are generally not used to treat breast cancer in premenopausal women, since the decrease in estrogen stimulates androgen production by the ovaries, which will counteract their effect. The most common side effects of aromatase inhibitors are joint stiffness or joint pain. Regular bone density testing is recommended for those taking aromatase inhibitor drugs.23-25
Alcohol Consumption
Alcohol consumption increases estrogen levels and breast cancer risk.26,27 A study28 proposed that increased aromatization may be a mechanism for feminization in some male alcoholics and for plasma estrogen level increases in postmenopausal women who consumed moderate levels of alcohol.
In conclusion, metabolic syndrome presents a unique challenge because of its multiple mechanisms. Addressing glucose metabolism with diet, exercise, and natural therapies is a mainstay of naturopathic treatment. In addition, several natural aromatase inhibitors may be useful for men with metabolic syndrome accompanied by increased estrogen and decreased testosterone concentrations. They may be particularly useful as an adjunct to testosterone therapy in these patients.
 Ron Steriti, ND, is researcher for Dr Jonathan V. Wright of the Tahoma Clinic and Meridian Valley Labs in Renton, Washington. He is a graduate of Southwest College of Naturopathic Medicine and has a PhD in electrical engineering from the University of Massachusetts at Lowell.
Ron Steriti, ND, is researcher for Dr Jonathan V. Wright of the Tahoma Clinic and Meridian Valley Labs in Renton, Washington. He is a graduate of Southwest College of Naturopathic Medicine and has a PhD in electrical engineering from the University of Massachusetts at Lowell.
Jonathan V. Wright is medical director and founder of the Tahoma Clinic and Meridian Valley Labs. He is a pioneer in holistic medicine and bioidentical hormone therapy.
References
1. Sirianni R, Chimento A, Malivindi R, Mazzitelli I, Andò S, Pezzi V. Insulin-like growth factor–I, regulating aromatase expression through steroidogenic factor 1, supports estrogen-dependent tumor Leydig cell proliferation. Cancer Res. 2007;67(17):8368-8377.
2. la Marca A, Morgante G, Palumbo M, Cianci A, Petraglia F, De Leo V. Insulin-lowering treatment reduces aromatase activity in response to follicle-stimulating hormone in women with polycystic ovary syndrome. Fertil Steril. 2002;78(6):1234-1239.
3. Cohen PG. Obesity in men: the hypogonadal–estrogen receptor relationship and its effect on glucose homeostasis. Med Hypotheses. 2008;70(2):358-360.
4. Cohen PG, Holbrook JM. Other pathways to the manifestations of the metabolic syndrome in males. Obes Res. 2004;12(9):1536.
5. Cohen PG. Aromatase, adiposity, aging and disease: the hypogonadal-metabolic-atherogenic-disease and aging connection. Med Hypotheses. 2001;56(6):702-708.
6. Leder BZ. Testosterone, estradiol and aromatase inhibitor therapy in elderly men. J Steroid Biochem Mol Biol. 2007;106(1-5):162-167.
7. Johnson RE, Murad MH. Gynecomastia: pathophysiology, evaluation, and management. Mayo Clin Proc. 2009;84(11):1010-1015.
8. Czajka-Oraniec I, Zgliczynski W, Kurylowicz A, Mikula M, Ostrowski J. Association between gynecomastia and aromatase (CYP19) polymorphisms. Eur J Endocrinol. 2008;158(5):721-727.
9. Advani A, Johnson SJ, Nicol MR, et al. Adult-onset hypogonadotropic hypogonadism caused by aberrant expression of aromatase in an adrenocortical adenocarcinoma. Endocr J. 2010;57(7):651-656.
10. Stratakis CA, Batista D, Sabnis G, Brodie A. Prepubertal gynaecomastia caused by medication or the aromatase excess syndrome. Clin Endocrinol (Oxf). 2004;61(6):779-780.
11. Shozu M, Sebastian S, Takayama K, et al. Estrogen excess associated with novel gain-of-function mutations affecting the aromatase gene. N Engl J Med. 2003;348(19):1855-1865.
12. Braunstein GD. Aromatase and gynecomastia. Endocr Relat Cancer. 1999;6(2):315-324.
13. Yazici G, Sayin M. Role of aromatase inhibitors in glucose intolerance of obese men. Med Hypotheses. 2008;71(4):612-613.
14. Chi TT. Sea cucumber extract and extracts of Cyperus and Curcuma. Int J Integr Oncol. 2008;2(1):7-15.
15. Chi TT. Myomin: natural aromatase inhibitor for estrogen-related conditions. Townsend Lett. 2009. http://www.thefreelibrary.com/Myomin%3a+natural+aromatase+inhibitor+for+estrogen-related+conditions.-a0213722869. Accessed September 7, 2010.
16. Chi TC. Effect of myomin in the expression of aromatase. Fertil Steril. 2010.
17. Brown GA, Vukovich MD, Martini ER, et al. Effects of androstenedione-herbal supplementation on serum sex hormone concentrations in 30- to 59-year-old men. Int J Vitam Nutr Res. 2001;71(5):293-301.
18. Brown GA, Vukovich MD, Martini ER, et al. Endocrine and lipid responses to chronic androstenediol-herbal supplementation in 30 to 58 year old men. J Am Coll Nutr. 2001;20(5):520-528.
19. Monteiro R, Azevedo I, Calhau C. Modulation of aromatase activity by diet polyphenolic compounds. J Agric Food Chem. 2006;54(10):3535-3540.
20. Balunas MJ, Su B, Brueggemeier RW, Kinghorn AD. Natural products as aromatase inhibitors. Anticancer Agents Med Chem. 2008;8(6):646-682.
21. Kraft JR. Detection of diabetes mellitus in situ (occult diabetes). Lab Med. 1975;6(2):20-22.
22. Zhao J, Dasmahapatra AK, Khan SI, Khan IA. Anti-aromatase activity of the constituents from damiana (Turnera diffusa). J Ethnopharmacol. 2008;120(3):387-393.
23. Gonnelli S, Petrioli R. “Aromatase inhibitors, efficacy and metabolic risk in the treatment of postmenopausal women with early breast cancer.” Clin Interv Aging. 2008;3(4):647-57.
24. Colozza M, Califano R, et al. “Aromatase inhibitors: a new reality for the adjuvant endocrine treatment of early-stage breast cancer in postmenopausal women.” Mini Rev Med Chem. 2008;8(6):564-74.
25. Cuzick J, Sestak I, et al. “Treatment-emergent endocrine symptoms and the risk of breast cancer recurrence: a retrospective analysis of the ATAC trial.” Lancet Oncol. 2008;9(12):1143-1148.
26. Turner RT, Sibonga JD. Effects of alcohol use and estrogen on bone. Alcohol Res Health. 2001;25(4):276-281.
27. Deandrea S, Talamini R, Foschi R. Alcohol and breast cancer risk defined by estrogen and progesterone receptor status: a case-control study. Cancer Epidemiol Biomarkers Prev. 2008;17(8):2025-2028.
28. Purohit V. Can alcohol promote aromatization of androgens to estrogens? a review. Alcohol
. 2000;22(3):123-127.