Erica Peirson, ND
Down syndrome (DS) is one of the most common congenital anomalies worldwide, affecting approximately 14 in 10 000 live births.1 The syndrome occurs as a result of extra genetic material, full or partial, from chromosome 21. This additional genetic material can lead to symptoms that include anatomic, immunologic, endocrine, metabolic, and neurological disorders characteristic of the phenotype. Every person with DS is a unique individual and may possess these characteristics to different degrees or not at all. There is a positive side to DS as well. Many parents of children with DS consider them a blessing. Individuals with DS are often very caring and sensitive and inspire others around them to be the same. Many adults with DS have jobs, get married, and contribute to society in countless ways.
Because of increasing accuracy and frequency of prenatal testing for DS, abortion rates among pregnant women who receive the diagnosis of DS for their fetus is around 67%.2 This number is lower than what has been previously reported at around 90%.2 Many parents choose this option owing to the financial and emotional toll that having a child with a disability can create. One of my goals in researching therapies for DS and in writing this article is to provide an alternative to parents who struggle with the decision of terminating a pregnancy as their only viable option. One might think that because DS is a genetic condition that nothing can be done to alter the biochemistry of someone with DS. Many conditions exist that have a genetic root cause, but they are still researched and treated. Such is the case for DS.
Down syndrome is the term used to describe the collection of clinical signs associated with the phenotype, which has several genetic causes. The most common cause is trisomy 21. This type affects roughly 96% of all patients with DS and involves a complete extra copy of chromosome 21.2 Down syndrome caused by translocation involves a partial duplication of chromosome 21 that is attached to another chromosome. Finally, there is mosaic DS, which affects about 1% of all patients with the syndrome.2 In this type, only a percentage of the patient’s cells contain the genetic anomaly. The rest of the patient’s cells are euploid. Much is still unknown about the true causes of DS symptoms. Although the Human Genome Project was completed in 2003, we still do not know the function of most of the genes on chromosome 21. In this article, I will present what is known and the ways in which we can alter the biochemistry of patients with DS.
Goals of Treatment
One of the main goals of treating patients with DS is in the area of cognition and neurology. Once life-threatening conditions such as heart defects and hypothyroidism are addressed, the following goals are sought: (1) Improving cognition leads to increased success with independent living, social interactions, and education. (2) Extending life expectancy includes reducing overall organ damage that occurs with DS and contributes to patients’ shorter life spans. (3) Preventing Alzheimer disease (AD) is paramount to helping patients with DS as they age.
Preventing Early-Onset AD
It has been estimated that almost all patients with DS older than 40 years show signs and symptoms of AD.3 Prevention of AD is key to helping patients with DS extend their lives without the distress that often accompanies AD. One of the key genes involved in AD is the amyloid precursor protein gene (APP),4 which is located on chromosome 21. Amyloid-β plaques are one of the main causes of AD. The extra copy of the APP gene that patients with DS have is implicated in their increased risk for acquiring early-onset AD. In addition, amyloid-β proteins contribute to degeneration of basal forebrain cholinergic neurons.5 These neurons are important for learning, memory, and attention. The amyloid protein plaques that develop in AD are present in all patients with DS who are older than 40 years.
As yet, there is no known treatment for AD. However, lithium has shown promising results in recent research in the area of AD prevention. Amyloid-β proteins are formed after cleavage of APP proteins. Lithium has been shown to decrease cleavage of APP proteins, reducing amyloid-β production and senile plaque formation.6 The subventricular zone of the brain is an area that retains neurogenic potential throughout life.7 Lithium was able to restore cell proliferation in these subventricular zone cells of mice, which are genetically and phenotypically similar to those of humans with DS.8 The use of lithium carbonate is associated with fewer adverse effects than lithium chloride, which has historically been prescribed.
Another promising candidate for preventing AD is docosahexaenoic acid. This important fatty acid seems to reduce the production of amyloid-β proteins.9
Curcumin has been used for centuries in Asian and Ayurvedic medicine. It is known to have anti-inflammatory and anticancer properties and can now be said to have antiamyloidogenic properties. It has the ability to reduce neurodegeneration, amyloid plaque formation. and inflammation, which are contributing factors to the development of AD.10
Oxidative Stress
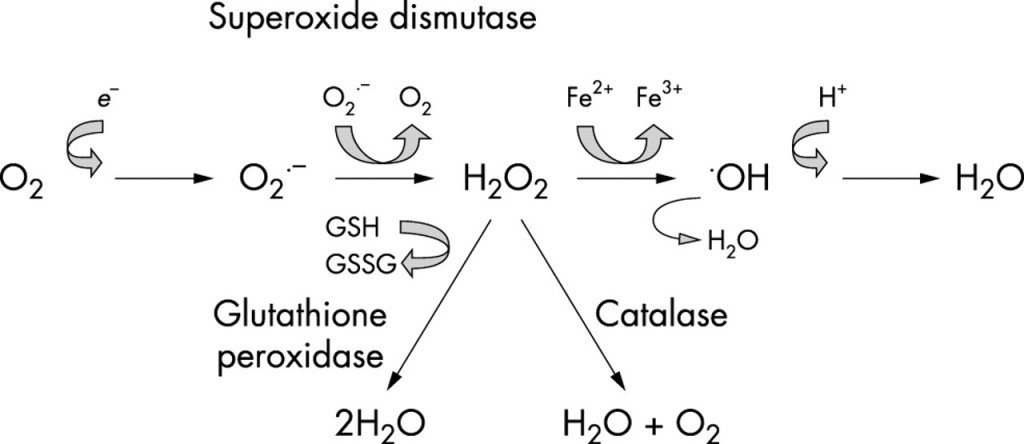
 The role of chromosomes is to code for proteins. Because patients with DS have extra chromosome material, they experience genetic overexpression of specific amino acids and polypeptides. One of the proteins that is overexpressed in DS is copper-zinc superoxide dismutase type 1 (SOD-1).11 The genes coding for this important antioxidant are found on chromosome 21 (21q21-22.3) in an area called the DS critical region. Case reports of partial trisomy 21 support the hypothesis that the genes in this region are involved in the phenotype of DS.12-14 Moreover, SOD-1 activity has been found to be increased by about 50% in all tissues of patients with DS.15
The role of chromosomes is to code for proteins. Because patients with DS have extra chromosome material, they experience genetic overexpression of specific amino acids and polypeptides. One of the proteins that is overexpressed in DS is copper-zinc superoxide dismutase type 1 (SOD-1).11 The genes coding for this important antioxidant are found on chromosome 21 (21q21-22.3) in an area called the DS critical region. Case reports of partial trisomy 21 support the hypothesis that the genes in this region are involved in the phenotype of DS.12-14 Moreover, SOD-1 activity has been found to be increased by about 50% in all tissues of patients with DS.15
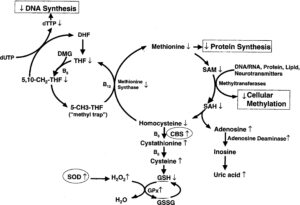
The overexpression of SOD would not be a problem if the genes that coded for glutathione and catalase were also on chromosome 21 in the DS critical region. However, the gene for glutathione synthetase is located on chromosome 20 (20q11.2), and the gene for catalase is on chromosome 11 (11p13). SOD-1 converts reactive oxygen species to hydrogen peroxide. This hydrogen peroxide is converted to water by glutathione peroxidase and catalase. This creates an imbalance in the SOD-glutathione system. When there is more SOD-1 than glutathione or catalase can keep up with, a buildup of harmful hydrogen peroxide subsists (Figure 1). The damage from hydrogen peroxide is made worse in the presence of iron via a Fenton reaction.
An imbalance between SOD and glutathione peroxidase activity has been proposed as a mechanism of the aging process.16,17 Hydrogen peroxide not only is a powerful oxidative agent but also stimulates apoptosis (programmed cell death).17 The excess accumulation of hydrogen peroxide is damaging to macromolecules such as DNA, protein, and lipids, leading to organelle damage (especially mitochondria18), cellular damage, tissue damage, and ultimately organ damage.
To complicate matters further, the gene for cystathionine β-synthase is also on chromosome 21 (21q22.3). This enzyme is involved in converting homocysteine to cysteine. An excess accumulation of cysteine results from the overexpression of cystathionine β-synthase, as well as a reduction in homocysteine levels (Figure 2).19 Once again, there is not enough glutathione synthetase to keep up with the excess cysteine. Unfortunately, excess levels of cysteine have been shown to have neurotoxic effects.20 The best means we have to increase glutathione levels in patients is N-acetylcysteine. However, increasing cysteine to raise glutathione levels does not work in patients with DS and is even detrimental. It seems that the rate-limiting step in glutathione synthesis rests at glutathione synthetase.
The reduction in homocysteine levels leads to a reduction in methionine levels and methionine synthase activity:
The decrease in the methionine synthase activity thus creates the well-established ‘methyl trap,’ as a consequence of the one-way kinetics of 5-MTHF synthesis [Figure 2]. More important, a decrease in methionine synthase activity reduces the conversion of 5-MTHF to tetrahydrofolate (THF), the metabolically active form of folate, required for de novo synthesis of nucleotides for RNA and DNA synthesis. Because of the methyl trap, an intracellular functional folate deficiency can exist in the presence of normal, or even elevated, serum folate and B12 levels.19(pp89-90)
Several supplements were studied for their effect on this imbalanced system.19 They are methyl B12, folinic acid, methionine, thymidine, and dimethylglycine. All were shown to be helpful, but the supplements most promising for increasing glutathione and methionine levels were methyl B12, folinic acid, and methionine.
Melatonin has shown some preliminary promise as an antioxidant agent used to raise glutathione levels. It has also been shown to raise SOD levels, so the use of it in DS is questionable and further studies need to be performed before its use can be warranted for this purpose.21 Liposomal glutathione used transdermally bypasses the use of cysteine as a precursor and shows promise for raising glutathione levels.22 A product that does not contain cysteine must be used for reasons discussed earlier. Intravenous glutathione is impractical given the daily and constant need for glutathione throughout the lifetime of a patient with DS. Other forms of administration are available, including nebulized glutathione, sublingual glutathione, and even glutathione suppositories. The absorption via these means has not been sufficiently studied, but their use has shown few adverse effects. Glutathione administered via nebulizer should not be used in patients with asthma because it can cause a worsening of symptoms.
Making strides to improve glutathione levels is key to altering the premature aging that is often seen in patients with DS. However, thus far, one single means of raising glutathione levels in patients with DS has not been found. Avoiding drugs and other agents that reduce glutathione levels is recommended. Acetaminophen should not be used routinely by patients with DS. The only time that acetaminophen can be used is before and after surgery because nonsteroidal anti-inflammatory drugs are avoided owing to their blood-thinning properties. Resveratrol is a powerful antioxidant that has been shown to reverse depletion of glutathione levels in mice induced with acetaminophen toxicity.23 As with melatonin, resveratrol has also been shown to increase SOD along with glutathione.24 Further studies are needed to test the use of resveratrol in the unique biochemical environment of patients with DS.
 Neurotransmitters
Neurotransmitters
A second imbalance exists in the excitatory and inhibitory impulses of the nervous system in patients with DS. It has been discovered that this imbalance is due to overactive γ-aminobutyric acid A (GABAA) receptors in the brain.25,26 GABA is the body’s main inhibitory neurotransmitter. Its role in moderating all other neurotransmitter activity is important. However, excessive GABA activity can lead to problems with memory, cognition, concentration, and decreased muscle tone. It is often prescribed for conditions such as anxiety, insomnia, and hypertonic muscles that benefit from inhibition of the nervous system. Researchers at the Texas Tech University Health Sciences Center, Lubbock, discovered that Ginkgo biloba has GABAA antagonist properties.27 This was exciting and useful news to those seeking to improve cognition in patients with DS. In addition, G biloba has been shown to help improve cognition in older patients with Alzheimer-type dementia.28 Ginkgo biloba is one of the most prescribed herbs among older populations for this reason. Given the connection between DS and the development of AD, G biloba is an important line of therapy when treating DS.
The dosage of G biloba is key. The recommended starting minimum dosage is 5 mg/kg 3 times a day. This dosage should be increased every 1 to 2 months until improvements in memory, speech, writing, socialization, and other areas of cognition are seen.
Acetylcholine is an important excitatory neurotransmitter of the peripheral nervous system. Patients with DS often experience low muscle tone, which is possibly due to decreased acetylcholine levels in the peripheral nervous system. Although its activity in the central nervous system is less significant, a decrease in the levels of acetylcholine has been implicated in cognitive delays in DS.29 Acetylcholine is produced from acetyl coenzyme A, from glucose metabolism, and from choline. The largest source of choline in the body comes from phosphatidylcholine, the major phospholipid in cell membranes of plants and animals. Increasing choline levels can be accomplished through oral supplementation with choline.30 Lecithin serves as an excellent source of choline as well.
It has long been accepted that patients with DS have low serotonin levels.31-33 Because melatonin is produced from serotonin, it follows that sleep disturbances in DS are likely due to altered serotonin levels. Behavioral problems have also been linked to low serotonin levels in patients with DS. Improving serotonin levels can be helpful for patients who experience sleep disturbances and mood and behavioral problems. 5-Hydroxytryptophan, the precursor to serotonin, is an ideal supplement for low serotonin levels in DS given its low rate of adverse effects compared with selective serotonin reuptake inhibitors. However, a 1971 study34 found that the administration of 5-hydroxytryptophan may cause spasms in infants. The dosage that led to these symptoms was 1.0 to 1.4 mg/kg in an infant aged 6 to 8 weeks. The patient did well once the dosage was adjusted.
Vitamins and Minerals
Vitamin therapy for patients with DS is important given their unique biochemical needs. Although no current formal studies have been performed to support the use of vitamins to improve cognition, many parents of children with DS report great strides in their child’s development after implementing vitamin therapy. A 1981 study35 showed an improvement in IQ and growth among children with DS after they had taken a vitamin-mineral supplement for 8 months. Many studies have been performed that report deficiencies of specific vitamins in patients with DS. The subsections that follow discuss zinc, vitamin A, and pyridoxine, which are often used to treat symptoms other than delayed cognition in DS. Many other supplements have been implicated, but a full report of them is outside the scope of this article. Although certain vitamins are reported to be low in patients with DS, each patient is unique. The use of high doses of vitamins should be considered only after vitamin-level test results are correlated with clinical signs and symptoms to create a catered treatment plan.
Zinc
Zinc deficiency has been implicated as a key factor in the increased rates of lymphopenia, skin conditions, hypogonadism, delayed growth, and upper respiratory tract infections seen in patients with DS.36,37 Plasma and urine levels of zinc have been shown to be low in children with DS.38 It can be postulated that the cause of low zinc levels is attributed to zinc being sequestered in its role as a cofactor for copper-zinc SOD that is overexpressed. However, studies linking these 2 factors have not been performed to date. Zinc deficiencies should be addressed regardless of their cause.
Vitamin A
Vitamin A is commonly used in the treatment of DS. The levels of vitamin A in patients with DS have been studied as far back as the 1970s.39,40 However, some evidence indicates that vitamin A deficiencies are seen in patients with DS,41 and other evidence indicates that no such deficiencies are seen.42 This is further proof that testing is important before beginning supplementation, especially with high-dose fat-soluble vitamins such as vitamin A. Signs of vitamin A deficiency include dry skin, dry hair, night blindness, broken fingernails, and decreased resistance to infections.
Pyridoxine
Pyridoxine (vitamin B6) metabolism in DS has been studied and was shown to improve the levels of 5-hydroxyindole, a metabolite of serotonin, as effectively as 5-hydroxytryptophan administration.43 The administered dosage of pyridoxine was 5 to 10 mg/kg. A more recent 2008 study44 revealed that the levels of oxalate, a marker for vitamin B6 deficiency, were elevated in the amniotic fluid of mothers carrying fetuses with DS. Because symptoms of vitamin B6 deficiency (seizures, poor growth, lymphopenia, and seborrheic dermatitis) are often seen in patients with DS, testing of xanthurenic acid levels, a more accepted biomarker for vitamin B6, is recommended.
l-carnitine
l-carnitine is an amino acid involved in transporting fatty acids into the mitochondria to be converted to acetyl coenzyme A, which is further converted to energy via the citric acid cycle. In patients with DS, l-carnitine can help improve muscle tone and overall energy. It also functions as an antioxidant, providing a protective effect against lipid peroxidation. Specifically, l-carnitine reduces oxidative damage due to hydrogen peroxide, which has previously been found to be elevated in patients with DS.45
Nutrition
Eating a nutrient-dense diet that includes lean protein, healthy fats, whole grains, and colorful antioxidant-rich fruits and vegetables is key for anyone wanting to live a long life. It is particularly important for patients with DS given their increased levels of oxidative stress. In addition, maintaining a healthy weight is vital to optimizing cardiovascular function and joint health. This can be a challenge for patients with DS if gross motor challenges prevent them from participating in exercise. Supplementing with vitamins that are found to be deficient can help with overall energy, neuromuscular function, and motivation to get up and move.
The incidence of celiac disease is approximately 3.2% to 7.0% in patients with DS compared with 1.0% in individuals without DS.46,47 Annual screening is important to rule out the development of gluten sensitivity or celiac disease. When possible, eliminating gluten completely from the diet of patients with DS is recommended. As well, gluten-derived and casein-derived opiate peptides inhibit cysteine absorption. Because cysteine is essential for the production of glutathione, removing gluten and casein from the diet of patients with DS can greatly help them optimize their glutathione levels.
Future Drug Targets
There is still a lot that is not understood about the etiology of DS. The amount of research in this area has increased greatly in recent years. Much of this is owing to the efforts of the Down Syndrome Research and Treatment Foundation. Their mission is to “stimulate biomedical research that will accelerate the development of treatments to significantly improve cognition, including memory, learning and speech, for individuals with Down syndrome in order that they: participate more successfully in school, lead more active and independent lives and avoid the early onset of Alzheimer’s Disease.”48 Their approach, although not naturopathic, is to seek drug targets. Much can be learned from the information they are gathering.
Researchers at The Johns Hopkins University, Baltimore, Maryland, have found that cerebellar precursor cells are less responsive to sonic hedgehog (Shh), a growth factor important in cell growth and development.49 They also discovered a drug called sonic agonist that mimics the effects of Shh and contributes to increased cerebellar growth in the DS mouse model. One injection of this drug at the time of birth resulted in improved cerebellar growth that translated to restored function of memory and learning on a hippocampus test in these mice.
The nervous system of the Caenorhabditis elegans worm is being used to study genes involved in the human nervous system by researchers at The University of Texas at Austin.50 Most human genes are shared with this worm, including roughly 80% of the genes from chromosome 21 in humans. The process of deleting or creating extra copies of genes in this worm is a simple process. Although it is not a good model for cognition and learning, it is an excellent model for learning which genes give rise to different types of neurons. Studying these neurons in this worm is much easier than studying the neurons of the human brain because the C elegans worm has exactly 302 neurons and the human brain has approximately 100 billion neurons. All the neurons of the C elegans worm have been mapped out. The researchers systematically knocked out each gene that corresponds to chromosome 21 in humans in a matter of 4 months. They discovered 9 genes involved in inhibitory signaling in the nervous system. The investigators have been able to create 226 transgenic C elegans worms having a different copy of each gene that correlates to chromosome 21 in humans. The efficacy of drugs for the treatment of neurodegenerative diseases such as AD and DS can be quickly ascertained owing to the short life span of these worms. As well, information gained from studying these worms can be passed on to researchers working with the DS mouse model, and further testing of potentially helpful drugs can be conducted.
Testing
As previously mentioned, testing is an important part of developing a treatment plan that is specific for each patient. Although patients with DS may have the same duplicate chromosome 21, they also have 22 other chromosomes that can affect their body’s need for supplementation. Several tests are helpful when working with patients with DS. One is a comprehensive functional nutrition assessment that covers antioxidants, vitamins, digestive support, essential fatty acids, amino acids, and minerals. Another is an oxidative stress test that provides SOD, glutathione, and lipid peroxidase levels, among others. Neurotransmitters can also be tested. This includes GABA, serotonin, and other neurotransmitters. It is helpful to acquire these test results before any treatment is begun to obtain baseline levels and to retest regularly to monitor treatment.
Prevention
Various studies51-53 from around the world have discovered a link between polymorphisms of the methylenetetrahydrofolate reductase gene (MTHFR) (A1298C and C677T) as a maternal risk factor for DS. Elevated homocysteine levels are more common in women who have a child with DS. Testing for this genetic defect in the mother can be helpful if future children are planned. It is especially important to test mothers who were younger than 30 years when their child with DS was conceived because this is not the typical age of mothers at risk for having a child with DS. Homocysteine levels should also be checked to ensure that the mother is not experiencing any other health issues associated with the MTHFR polymorphism. It is important to counsel the parents about the risk for having future children with DS if the MTHFR gene defects are found and are not addressed. Screening the child with DS for the MTHFR defect is helpful as well because the genetic polymorphism can be passed on to the child, compromising his or her health beyond existing issues from DS. If one or both MTHFR defects are found, they need to be addressed in addition to the imbalances discussed earlier.
Keep Up With Research
Assisting patients with DS is a complicated endeavor given that so many organ systems are affected. Much research has been performed to understand the biochemistry that causes the symptoms of DS and the potential treatments that can help optimize health and cognition. More research is needed to fully understand the syndrome as a whole. The current standard of care for patients with DS includes little to none of the information offered in this article. Allopathic medical pediatricians need to catch up with the research that is available to better aid their patients with DS.
 Erica Peirson, ND is a graduate of National College of Natural Medicine (Portland, Oregon). She is passionate about helping children with Down Syndrome reach their fullest potential, and lectures on the topic as well as treats patients privately in her practice. Her journey of studying the biochemistry of Down Syndrome began 4 years ago when her son was born with Mosaic Down Syndrome. Her practice also focuses on weight loss, thyroid conditions, fatigue, insomnia, and bioidentical hormone replacement therapy. When not seeing patients, she teaches Cell Biology and Anatomy & Physiology at Portland Community College.
Erica Peirson, ND is a graduate of National College of Natural Medicine (Portland, Oregon). She is passionate about helping children with Down Syndrome reach their fullest potential, and lectures on the topic as well as treats patients privately in her practice. Her journey of studying the biochemistry of Down Syndrome began 4 years ago when her son was born with Mosaic Down Syndrome. Her practice also focuses on weight loss, thyroid conditions, fatigue, insomnia, and bioidentical hormone replacement therapy. When not seeing patients, she teaches Cell Biology and Anatomy & Physiology at Portland Community College.
References
Parker SE, Mai CT, Canfield MA, et al; National Birth Defects Prevention Network. Updated national birth
prevalence estimates for selected birth defects in the United States, 2004-2006. Birth Defects Res A Clin Mol Teratol. 2010;88(12):1008-1016.
National Down Syndrome Society. What causes Down
syndrome? http://www.ndss.org/en/About-Down-Syndrome/What-Causes-Down-Syndrome. Accessed August 6, 2012.
Zis P, Dickinson M, Shende S, Walker Z, Strydom A. Oxidative stress and memory decline in adults with Down syndrome: longitudinal study [published online May 4, 2012]. J Alzheimers Dis. doi:10.3233/JAD-2012-120073. Medline:22561328
Salehi A, Delcroix JD, Belichenko PV, et al. Increased App expression in a mouse model of Down’s syndrome disrupts NGF transport and causes cholinergic neuron degeneration. Neuron. 2006;51(1):29-42.
Patel AN, Jhamandas JH. Neuronal receptors as targets for the action of amyloid-beta protein (Aβ) in the brain. Expert Rev Mol Med. 2012;14:e2. http://journals.cambridge.org/action/displayAbstract?fromPage=online&aid=8475686. Accessed July 16, 2012.
Leroy K, Ando K, Heraud C, et al. Lithium treatment arrests the development of neurofibrillary tangles in mutant tau transgenic mice with advanced neurofibrillary pathology.
J Alzheimers Dis. 2010;19:705-719.
Doetsch F, Caillé I, Lim DA, García-Verdugo JM, Alvarez-Buylla A. Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell. 1999;97(6):703-716.
Bianchi P, Ciani E, Contestabile A, Guidi S, Bartesaghi
R. Lithium restores neurogenesis in the subventricular zone of the Ts65Dn mouse, a model for Down syndrome. Brain Pathol. 2010;20(1):106-118.
Cole GM, Frautschy SA. DHA may prevent age-related dementia. J Nutr. 2010;140(4):869-874.
Begum AN, Jones MR, Lim GP, et al. Curcumin structure-function, bioavailability, and efficacy in models of neuroinflammation and Alzheimer’s disease. J Pharmacol Exp Ther. 2008;326(1):196-208.
Annerén G, Edman B. Down syndrome: a gene dosage disease caused by trisomy of genes within a small segment of the long arm of chromosome 21, exemplified by the study of effects from the superoxide-dismutase type 1 (SOD-1) gene. APMIS Suppl. 1993;40:71-79.
Nadal M, Moreno S, Pritchard M, Preciado MA, Estivill X, Ramos-Arroyo MA. Down syndrome: characterisation of a case with partial trisomy of chromosome 21 owing to a paternal balanced translocation (15;21)(q26;q22.1) by FISH. J Med Genet. 1997;34:50-54.
Korenberg JR, Chen XN, Schipper R, et al. Down syndrome phenotypes: the consequences of chromosomal imbalance. Proc Natl Acad Sci U S A. 1994;91(11):4997-5001.
Delabar JM, Theophile D, Rahmani Z, et al. Molecular mapping of twenty-four features of Down syndrome on chromosome 21. Eur J Hum Genet. 1993;1:114-124.
De La Torre R, Casado A, López-Fernández E, Carrascosa D, Ramirez V, Sáez J. Overexpression of copper-zinc superoxide dismutase in trisomy 21. Experientia. 1991;52:871-873.
Bar-Peled O, Korkotian E, Segal M, Groner Y. Constitutive overexpression of Cu/Zn dismutase exacerbates kainic acid–induced apoptosis of transgenic–Cu/Zn superoxide dismutase neurons. Proc Natl Acad Sci U S A. 1996;93(16):8530-8535.
de Haan JB, Cristiano F, Iannello R, Bladier C, Kelner MJ, Kola I. Elevation of the ratio of Cu/Zn–superoxide dismutase to glutathione peroxidase activity induces features of cellular senescence and this effect is mediated by hydrogen peroxide. Hum Mol Genet. 1996;5(2):283-292.
Pagano G, Castello G. Oxidative stress and mitochondrial dysfunction in Down syndrome. Adv Exp Med Biol. 2012;724:291-299.
Pogribna M, Melnyk S, Pogribny I, Chango A, Yi P, James SJ. Homocysteine metabolism in children with Down syndrome: in vitro modulation. Am J Hum Genet. 2001;69(1):88-95.
Janáky R, Varga V, Hermann A, Saransaari P, Oja SS. Mechanisms of l-cysteine neurotoxicity. Neurochem Res. 2000;25(9-10):1397-1405.
Pandi-Perumal SR, Bahammam AS, Brown GM, et al. Melatonin antioxidative defense: therapeutical implications for aging and neurodegenerative processes [published online June 28, 2012]. Neurotox Res. doi:10.1007/s12640-012-9337-4. Medline:22739839
Zeevalk GD, Bernard LP, Guilford FT. Liposomal-glutathione provides maintenance of intracellular glutathione and neuroprotection in mesencephalic neuronal cells. Neurochem Res. 2010;35(10):1575-1587.
Sener G, Toklu HZ, Sehirli AO, Velioğlu-Oğünç A, Cetinel S, Gedik N. Protective effects of resveratrol against acetaminophen-induced toxicity in mice. Hepatol Res. 2006;35(1):62-68.
Spanier G, Xu H, Xia N, et al. Resveratrol reduces endothelial oxidative stress by modulating the gene expression of superoxide dismutase 1 (SOD1), glutathione peroxidase 1 (GPx1) and NADPH oxidase subunit (Nox4). J Physiol Pharmacol. 2009;60(suppl 4):111-116.
Kleschevnikov AM, Belichenko PV, Villar AJ, Epstein CJ, Malenka RC, Mobley WC. Hippocampal long-term potentiation suppressed by increased inhibition in the Ts65Dn mouse, a genetic model of Down syndrome. J Neurosci. 2004;24(37):8153-8160.
Belichenko PV, Kleschevnikov AM, Masliah E, et al. Excitatory-inhibitory relationship in the fascia dentata
in the Ts65Dn mouse model of Down syndrome.
J Comp Neurol. 2009;512(4):453-466.
Kiewert C, Kumar V, Hildmann O, et al. Role of GABAergic antagonism in the neuroprotective effects of bilobalide. Brain Res. 2007;1128(1):70-78.
Mazza M, Capuano A, Bria P, Mazza S. Ginkgo biloba and donepezil: a comparison in the treatment of Alzheimer’s dementia in a randomized placebo-controlled double-blind study. Eur J Neurol. 2006;13(9):981-985.
Chang Q, Gold PE. Age-related changes in memory and in acetylcholine functions in the hippocampus in the Ts65Dn mouse, a model of Down syndrome. Neurobiol Learn Mem. 2008;89(2):167-177.
Babb SM, Ke Y, Lange N, Kaufman MJ, Renshaw PF, Cohen BM. Oral choline increases choline metabolites in human brain. Psychiatry Res. 2004;130(1):1-9.
Lott IT, Murphy DL, Chase TN. Down’s syndrome: central monoamine turnover in patients with diminished platelet serotonin. Neurology. 1972;22(9):967-972.
Kamoun P, Lafourcade G, Jerome H. Catabolism of serotonin in Down’s syndrome (trisomy 21). Biomedicine. 1974;21(10):426-428.
Airaksinen EM. Platelet-rich plasma 5-hydroxytryptamine, urinary 5-hydroxyindoleacetic acid and tryptophan ingestion in Mongols. J Ment Defic Res. 1971;15 pt 4(0):244-256.
34. Coleman M. Infantile spasms associated with 5-hydroxytryptophan administration in patients with Down syndrome. Neurology. 1971;21(9):911-919.
35. Harrell RF, Capp RH, Davis DR, Peerless J, Ravitz LR. Can nutritional supplements help mentally retarded children? an exploratory study. Proc Natl Acad Sci U S A. 1981;78(1):574-578.
36. Ram G, Chinen J. Infections and immunodeficiency in Down syndrome. Clin Exp Immunol. 2011;164(1):9-16.
37. Prasad AS. Discovery of human zinc deficiency: 50 years later. J Trace Elem Med Biol. 2012;26(2-3):66-69.
38. Lima AS, Cardoso BR, Cozzolino SF. Nutritional status of zinc in children with Down syndrome. Biol Trace Elem Res. 2010;133(1):20-28.
39. Barden HS. Vitamin A and carotene values of institutionalised mentally retarded subjects with and without Down’s syndrome. J Ment Defic Res. 1977;21(1):63-74.
40. Palmer S. Influence of vitamin A nutriture on the immune response: findings in children with Down’s syndrome. Int J Vitam Nutr Res. 1978;48(2):188-216.
41. Matin MA, Sylvester PE, Edwards D, Dickerson JW. Vitamin and zinc status in Down syndrome. J Ment Defic Res. 1981;25(pt 2):121-126.
42. Erics M, Balci S, Atakan N. Dermatological manifestations of 71 children admitted to a clinical genetic unit. Clin Genet. 1996;50(5):317-320.
43. Pueschel SM, Reed RB, Cronk CE, Goldstein BI. 5-Hydroxytryptophan and pyridoxine: their effects in young children with Down’s syndrome. Am J Dis Child. 1980;134(9):838-844.
44. Baggot PJ, Eliseo AJY, DeNicola NG, Kalamarides JA, Shoemaker JD. Pyridoxine-related metabolite concentrations in normal and Down syndrome amniotic fluid. Fetal Diagn Ther. 2008;23(4):254-257.
45. Yu J, Ye J, Liu X, Han Y, Wang C. Protective effect of l-carnitine against H2O2-induced neurotoxicity in neuroblastoma (SH-SY5Y) cells. Neurol Res. 2011;33(7):708-716.
46. Zachor DA, Mroczek-Musulman E, Brown P. Prevalence of celiac disease in Down syndrome in the United States. J Pediatr Gastroenterol Nutr. 2000;31(3):275-279.
47. Mackey J, Treem WR, Worley G, et al. Frequency of celiac disease in individuals with Down syndrome in the United States. Clin Pediatr. 2001;40(5):249-252.
48. Down Syndrome Research and Treatment Foundation. Our mission. http://www.dsrtf.org/page.aspx?pid=348. Accessed July 4, 2012.
49. Currier DG, Polk RC, Reeves RH. A sonic hedgehog (Shh) response deficit in trisomic cells may be a common denominator for multiple features of Down syndrome. Prog Brain Res. 2012;197:223-236.
50. Down Syndrome Research and Treatment Foundation. DSRTF’s webinars: Dr. Jon Pierce-Shimomura, University of Texas at Austin, Down Syndrome Cognition Research, Tuesday, May 22, 2012. http://www.dsrtf.org/page.aspx?pid=551. Accessed May 22, 2012.
51. Vraneković J, Babić Bozović I, Starcević Cizmarević N, et al. Functional inference of methylenetetrahydrofolate reductase gene polymorphisms on enzyme stability as a potential risk factor for Down syndrome in Croatia. Dis Markers. 2010;28(5):293-298.
52. Cyril C, Rai P, Chandra N, Gopinath PM, Satyamoorthy K. MTHFR gene variants C677T, A1298C and association with Down syndrome: a case-control study from South India. Indian J Hum Genet. 2009;15(2):60-64.
53. Biselli JM, Goloni-Bertollo EM, Zampieri BL, Haddad R, Eberlin MN, Pavarino-Bertelli EC. Genetic polymorphisms involved in folate metabolism and elevated plasma concentrations of homocysteine: maternal risk factors for Down syndrome in Brazil. Genet Mol Res. 2008;7(1):33-42.