Achieving Balance in the Thyroid
Alan Christianson, NMD
I loved naturopathic medical school. Learning new ideas and exotic treatments was like exploring a strange new country.
Perhaps, it was being in the first class of a brand-new school. Maybe, it was the spirit of curiosity among my classmates. For whatever reason, I was compelled to experience it all firsthand. Supplements, herbs, and homeopathic agents were all readily available. The effects from most of them were subtle at best. A few acted as memorable emetics.
Natural dessicated thyroid was a notable exception. I had been led to believe that it was a sure fix for fatigue. Figuring I would do well with a boost, I tried a ½-grain tablet. The effects were anything but subtle. That single dose produced no less than 12 hours of palpitations and sheer terror. I was left with the indelible impression that this was not something to be taken lightly.
At this time, in the mid 1990s, ideas on thyroid treatment by Broda Barnes and Denis Wilson were still new and creating tremendous interest.1 Many authors have since written about alternative treatment of thyroid disease, but none have strayed far from the model that Barnes produced.
Among the radical ideas was the proposition that a patient’s basal body temperature (BBT) could establish a diagnosis of hypothyroidism and determine optimal thyroid dosing. A morning axillary BBT below 97.8°F diagnosed hypothyroidism, and thyroid dosing could be safely increased so long as BBT did not surpass 98.2°F.1
A concomitant opinion was thyroid serologic test results, including thyrotropin, triiodothyronine (T3), and thyroxine (T4) levels, could be disregarded. Patients with normal levels could still be diagnosed as having hypothyroidism. Furthermore, patients who manifested hyperthyroid levels could be maintained or even escalated on thyroid treatment.
The model by Barnes1 was essentially 2-fold:
(1) BBT and symptoms can diagnose thyroid status.
(2) Thyroid serologic test results can be disregarded.
This broadening of diagnostic criteria inversely paralleled the narrowing of criteria in conventional medicine. Improvement in thyrotropin testing replaced symptom diagnosis and clinical judgment and left serologic testing as the sole arbiter of who has hypothyroidism and how much thyroid should be taken.
In the conventional diagnostic model, hypothyroidism exists only with a high thyrotropin level and a low T4 level. Hypothyroid symptoms and signs of Hashimoto thyroiditis may be present, but they cannot influence diagnosis in the case of normal serologic test results. Specifically, current definitions of hypothyroidism require repeated thyrotropin levels above 7.0 mIU/L, plus repeated total T4 or free T4 levels below local reference ranges, typically 5.6 µg/dL and 0.7 ng/dL, respectively (to convert total T4 level to nanomoles per liter and free T4 level to picomoles per liter, multiply by 12.871).2
The conventional model can be briefly summarized by the following tenets:
- Hypothyroidism can exist only with abnormal thyroid serologic test results.
- Patients with normal serologic test results cannot benefit from thyroid therapy.
The fact is that a large body of medically savvy “worried well” adult patients exist. Many have ongoing symptoms like muscle pain, fatigue, depression, and weight gain. Some of these patients have early thyroid disease, and some do not. Many suspect thyroid disease as the cause of their symptoms and are put off when told otherwise.
Just as it was getting harder to obtain thyroid treatment from a conventional physician, it was getting easier to obtain from an alternative one.
The resulting belief system in natural medicine was that almost any symptom could be reversed by the skillful administration of thyroid hormone. When this did not pan out, it was always the physician, not the model, who was to blame.
I saw numerous patients with fibromyalgia, chronic fatigue, and unexplained weight gain completely reverse their conditions with thyroid treatment. However, after my first few years of practice, I could no longer deny the apparent truth that the Barnes thyroid model helped many patients but had its shortcomings. Many other cases existed in which the same symptoms were present along with the same “low” BBT, yet thyroid therapy did not help these patients at all. In some of them, it caused troublesome adverse effects.
“Sonja” is a memorable example. She complained of unresolved fatigue, irritable bowel syndrome, and an inability to lose 50 lb gained with her last pregnancy. A friend who had experienced dramatic relief from similar symptoms had referred her. Sonja hoped for the same.
Her thyroid panel, consisting of thyrotropin, free T3, free T4, and thyroid antibodies, showed no abnormalities, with a thyrotropin level of 1.7 mIU/L. Her thyroid examination was unremarkable, as were the rest of her physical findings. Other test results showed casein and albumin intolerance and slightly elevated lipid levels. Cortisol, dehydroepiandrosterone, and ovarian hormone levels were reasonably within the normal range. Her blood pressure was normal, and she reported no symptoms of adrenal dysfunction. At a follow-up visit, her requested axillary BBT showed a mean of 96.1°F. Given my training in the Barnes model and experience up to that point, I expected her to respond well to natural dessicated thyroid.
Within a few days of starting a very conservative dosage of natural dessicated thyroid (¼ grain), she called back in a panic. Her heart was racing, and she had tremors and anxiety. She had not slept much at all for the past several days. She was upset with me, and so was I. She fit the Barnes profile, had no adrenal or cardiac contraindications to treatment, but did very poorly with it.
Now 14 years later, almost every week I meet another new patient who experienced something similar from another physician’s guidance. Therefore, I have developed the following objections to the Barnes model of hypothyroidism:
- BBT is too nonspecific to determine who needs thyroid treatment or to determine what is the correct thyroid dosing.
- Hyperthyroid serologic test results cannot be safely ignored
Shortcomings of BBT Testing
In his book Hypothyroidism: The Unsuspected Illness, Barnes states:
“A temperature below 97.8 indicates hypothyroidism…there is no risk of excessive thyroid dosage as long as basal temperature does not exceed 98.2.”1(p46) He cites no external data on the origins of this range but largely refers to his personal observations:
“Thus, it seemed that axillary, or underarm, temperature might serve as a simple guide to determining low thyroid function and the need for thyroid therapy. And over the past 30 years, it has served as such. In that time, based on many thousands of readings, it has been established that normal values for underarm temperature are in the range of 97.8-98.2 degrees Fahrenheit.”1(p46)
In 2002, a comprehensive review of articles from 1935 to 1999 on normal human body temperature was published.3 This meta-analysis showed that the range of normal oral temperatures was 91.8 to 100.6°F for women and 96.2 to 99.9°F for men. A subset of studies looking specifically at axillary temperatures but not factoring in sex showed a normal range of 95.6 to 98.6°F. Authors of the meta-analysis concluded: “The ranges of normal body temperature need to be adjusted, especially for the lower values. When assessing body temperature it is important to take place of measurement and gender into consideration.”3(p122)
If normal female BBT varies by 8.8°F, the Barnes model variation of only 0.4°F would be expected to yield an overwhelming number of abnormal readings, especially on the lower side.
Along with being too nonspecific to diagnose hypothyroidism, BBT cannot be safely relied on to prevent hyperthyroid adverse effects of thyroid therapy. The partial truth is that BBT elevates well above normal in cases of thyroid storm.4 Short of this crisis state, hyperthyroidism does not consistently or proportionately raise BBT.5,6 Therefore, BBT is not a reliable indicator of hyperthyroidism.
In summary, the BBT range that Barnes calls normal is far too narrow and on the high end. To state that one is safe from thyroid overdose with BBT below 98.2°F is erroneous.
Dangers of Too Much
As thyrotropin tests became more established, followers of Barnes were forced to confront the fact that thyroid dosing based on BBT often left the patient with a hyperthyroid thyrotropin level. Some have argued that a suppressed thyrotropin level is safe to ignore:
“Patients’ thyroid glands remain robust and patients don’t suffer ill effects unless they have symptoms of hyperthyroidism or their axillary basal temperature rises above 98.2.”7(p177)
Another similar argument originates from the alternative literature:
“The patient’s suppressed thyrotropin, then, does not indicate that the patient is over-treated with thyroid hormone; instead, it indicates that the patent’s low total thyroid hormone pool will finally rise to potentially adequate levels.”8
Substantial bodies of data tracking patients with suppressed thyrotropin levels show that morbidity and mortality increase even in the absence of obvious hyperthyroid symptoms.
These data come from the following 3 groups: (1) Patients found from screening to have subclinical hyperthyroidism, meaning abnormally low thyrotropin level, normal T3 and T4 levels, and no obvious hyperthyroid symptoms. (2) Patients with refractory endogenous hyperthyroidism, typically Graves disease. (3) Patients with intentional endogenous hyperthyroidism, generally to lower the risk of thyroid cancer recurrence.
Specific problems that become apparent with hyperthyroidism are higher rates of atrial fibrillation,9,10 stroke,11 osteoporosis,12 dementia,13 and mortality.14 Low thyrotropin levels independent of other markers of thyroid function have been shown to be a risk factor for osteoporosis in older persons.12
Shortcomings of the Conventional Model
Although the Barnes model of treatment is too broad, the conventional model is too restrictive. A case illustration follows.
“Joan” was a 43-year-old mother. Several months previously, she realized that she was depressed for no reason. Her workouts became harder to complete. She also noticed her periods becoming heavier and normal premenstrual syndrome symptoms getting worse. During this time, Joan saw that her hair was getting thinner. Without changes in her diet, she was rapidly gaining weight.
Joan’s mother reminded her about having the same experience in her early 40s. Joan’s mother was diagnosed as having hypothyroidism. Before seeing me, both her gynecologist and her family practice physician tested her thyroid levels and told her they were normal.
Joan’s examination and subsequent ultrasonographic findings were positive for thyroid nodules. Her blood tests showed a high normal thyrotropin level and positive thyroid antibodies. After 3 months of thyroid therapy, her mood had definitely improved, and her last few periods were easier. Her hair loss stopped, and she lost about 12 lb.
Joan is typical of many patients we have all seen. However, none of them would have undergone treatment based on conventional guidelines.
My main objections to the conventional model of thyroid disease are as follows:
- Thyroid symptoms can emerge, despite normal thyroid serologic test results.
- Some patients with thyroid symptoms and normal serologic test results can benefit from thyroid therapy.
- Some patients with thyroid symptoms and normal serologic test results are at higher medical risk without thyroid therapy.
- “Normal” thyroid serologic test results are not normal.
Symptoms Can Precede Changes in Serologic Test Results
In places like America that have salt iodination, hypothyroidism is caused primarily by Hashimoto thyroiditis.15 This is an immunologic assault against the thyroid tissue, led primarily by lymphocytes. Clear signs of the disease can be documented well before hypothyroid serologic test results manifest.15 The early stages of this disease can be documented by blood tests for antithyroid antibodies, palpable thyroid changes on physical examination, ultrasonographic findings, or tissue biopsy.16 Note that the absence of serum antibodies does not rule out Hashimoto thyroiditis.17
Because Hashimoto thyroiditis is ultimately a histological diagnosis, no absolute rule-out tests exist short of complete microscopic examination on an excised thyroid gland.
Early stages of the disease process can exist without changes in thyrotropin level but can cause hypothyroid symptoms, as illustrated in a recent study.18 In total, 426 women undergoing thyroid surgery for benign thyroid disease were questioned about hypothyroid symptoms and screened for thyroid hormone blood levels (Table 1). On histological evaluation of their thyroid glands, many were found to have Hashimoto thyroiditis. Regardless of thyroid hormone blood levels, histological evidence of Hashimoto thyroiditis was significantly associated with hypothyroid symptoms. The authors concluded that the autoimmune process was clearly able to cause hypothyroid symptoms even without serum hypothyroidism.
Thyroid therapy at an early stage of Hashimoto thyroiditis can decrease symptoms.19 Furthermore, severity of disease progression in these patients can be reduced with thyroid treatment.19

Patients With Thyroid Disease and Normal Serologic Test Results Can Benefit From Thyroid Treatment Medical Risks for Those Untreated
Along with improvement of hypothyroid symptoms, individuals with early thyroid disease have numerous medical risks that can improve with thyroid treatment.
Even without positive antithyroid antibodies and serum hypothyroidism, patients with Hashimoto thyroiditis have higher risks for heart disease.20,21
It has been argued that patients with Hashimoto thyroiditis whose thyrotropin level is above 2.0 mIU/L have an increased risk of developing thyroid cancer.22
In a representative study,23 postsurgical outcomes in 843 patients undergoing thyroid surgery to investigate for possible thyroid cancer were compared relative to their presurgical thyrotropin levels. Those with thyrotropin levels above 1.40 mIU/L represented 87% of all cases found to be cancerous.
Normal vs Optimal
For diagnosis, the conventional model relies exclusively on normal ranges to determine when to initiate thyroid therapy and when satisfactory dosing is achieved.
However, a strong case can be made that what we consider normal is biased toward those with thyroid disease and away from the healthy population. Most patients undergoing thyroid testing do so because they are known to have thyroid disease and are being monitored to assure proper dosing of thyroid therapy. Another sizable group of patients are being screened because of symptoms suspicious of hypothyroidism.
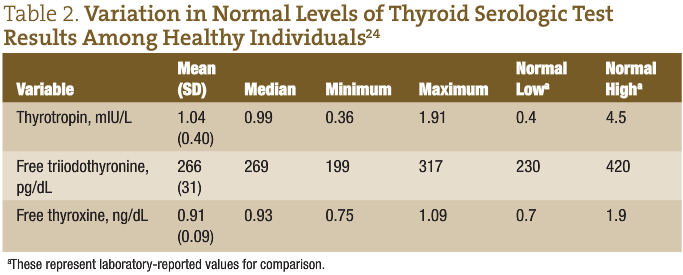
In a 2008 study,24 the mean thyrotropin, free T3, and free T4 levels of healthy individuals without thyroid disease were compared with current normal ranges (Table 2). Individuals chosen for the study were free of thyroid disease and had no chronic disease, such as diabetes mellitus, hypertension, coronary artery disease, or anemia. Persons were excluded in the event of pregnancy, a history of substance abuse, any medication use (including oral contraceptives), or any major medical illness (such as other endocrine or autoimmune disease).
This population showed a much more narrow range of thyroid serologic test result levels than typically observed. In healthy populations, thyrotropin, free T3, and free T4 levels fall at the lower end of the normal range.
Emerging Model and Terminology
Clearly, some patients do not have serum hypothyroidism but have hypothyroid symptoms that can be successfully treated with thyroid therapy. This condition has variously been called hypothyroidism, subclinical hypothyroidism, hypothyroidism type 2, or early Hashimoto thyroiditis.
To simply call this hypothyroidism is incorrect because hypothyroidism is currently defined by serologic test results. Subclinical hypothyroidism is both semantically incorrect and already in use for a distinct phenomenon. We cannot logically say “subclinical” when we are referring to symptoms, which are a clinical manifestation. Subclinical hypothyroidism is a phenomenon defined as a slightly elevated thyrotropin level, normal T4 level, and lack of clinical symptoms.25
Hypothyroidism type 2 is not a medically consistent term because its pathogenesis and treatment are not distinct from those of standard hypothyroidism.
Although cumbersome, early Hashimoto thyroiditis is probably closest to being correct. However, it too does not fit because early Hashimoto thyroiditis can exist without symptoms or future progression.26
To be consistent and brief, I call this condition clinical hypothyroid syndrome (CHS):
clinical because clinical data, including symptoms and physical examination findings, are pivotal to the diagnosis; hypothyroid because the pathogenesis, symptoms, and treatment overlap with those of hypothyroidism; and syndrome because it does not represent a single pathological finding but rather a defined constellation of findings and symptoms.
CHS Diagnosis
Over the ensuing years, I have formulated a diagnostic model for CHS. It works well to predict which patients without serum hypothyroidism will likely respond well to thyroid therapy and which patients will not.
Our initial points of contact with patients are their key symptoms. Certain symptoms should lead the clinician to suspect thyroid disease. Many books cite exhaustive lists of possible thyroid symptoms. These can be useful to be aware of, but many listed symptoms can be present at some point within health and within other disease processes.
The Colorado Thyroid Disease Prevalence Study27 represents our best scholarly examination of which symptoms best predict thyroid disease. In 1995, more than 25 000 individuals participated in health fairs in Colorado to obtain basic medical screening for vision, blood pressure, colon cancer, and skin cancer. Participants also had their serum thyrotropin level measured and were administered a thyroid symptom survey.
Blood thyroid levels were categorized as follows: (1) normal (thyrotropin level, 0.3-5.1 mIU/L), (2) subclinical hypothyroid (thyrotropin level, >5.1 mIU/L; normal T4 level), (3) hypothyroid (thyrotropin level, >5.1 mIU/L; low T4 level), and (4) hyperthyroid (thyrotropin level, <0.01 mIU/L).
Of many symptoms surveyed, those listed in Table 3 served as the best predictors of which patients would have significantly abnormal blood thyroid levels.
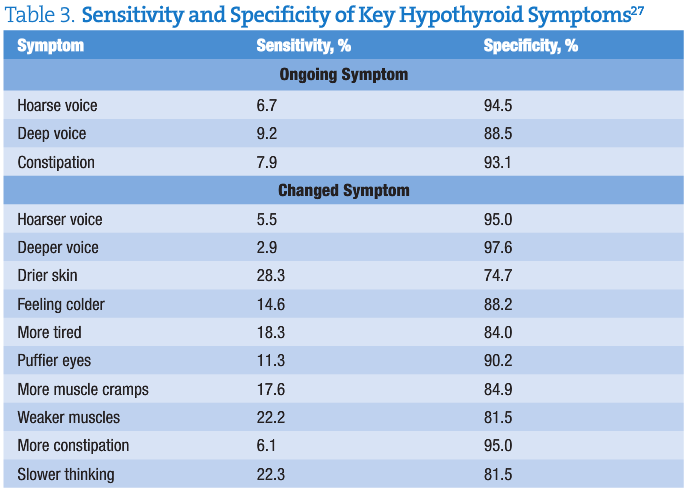
If a patient is seen with thyroid-specific symptoms, we should first perform a thorough physical examination and general testing to rule out other disorders. These include but are not limited to Addison disease, anemia, cardiovascular disease, diabetes mellitus, infectious mononucleosis, malignant neoplasm, medication adverse effects, rheumatologic disease, and sleep apnea.A notable observation in Table 3 is that no symptom has a very high sensitivity. Thyroid disease cannot be strongly ruled out in the absence of symptoms. Furthermore, the Colorado Thyroid Disease Prevalence Study27 found that most persons with newly discovered thyroid disease had fewer than 4 symptoms, which differed across patients. As in Table 3, it is useful to distinguish changed symptoms (defined as emerging in the past 6 months) from ongoing symptoms.
After ruling out other causes, CHS can be said to be present when all of the following 3 criteria exist:
- Recent onset of hypothyroid-specific symptoms (including fatigue, dry skin, cold intolerance, muscular weakness, dysphagia, chronic irritability, nervousness, and mood swings).
- One or more objective signs of Hashimoto thyroiditis (positive thyroid antibodies or thyroid structure abnormalities as evidenced by examination, ultrasonography, or biopsy).
- Suboptimal thyrotropin serologic test results (thyrotropin level, >2.0 mIU/L).
In closing, many patients with hypothyroid symptoms and normal thyroid serologic test results exist. If they receive thyroid therapy based on BBT and symptoms only, some will develop adverse effects and morbidity.
Conventional guidelines exclude this population from consideration of thyroid treatment. However, the literature clearly shows that a subset of them have early thyroid disease as the cause of their symptoms and may benefit from treatment, including thyroid therapy.
Alan Christianson, NMD, was a member of the first graduating class of Southwest College of Naturopathic Medicine and Health Sciences, Tempe, Arizona. His practice focus is thyroid disease and natural endocrinology. He has completed The Complete Idiot’s Guide to Thyroid Disease (Penguin Press, 2011) and is rewriting chapters on thyroid disease for the Textbook of Natural Medicine (Pizzorno).
References
1. Barnes B. Hypothyroidism: The Unsuspected Illness. New York, NY: HarperCollins Publishers; 1976:46.
2. Wiersinga WM. Adult hypothyroidism and myxedema coma. In: DeGroot LJ, Jameson JL, eds. Endocrinology. 5th ed. Philadelphia, PA: WB Saunders Company; 2004. Chapter.107.
3. Sund-Levander M, Forsberg C, Wahren L, et al. Normal oral, rectal, tympanic and axillary body temperature in adult men and women: a systematic literature review. Scand J Caring Sci. 2002;16(2):122-128.
4. Erkintalo M, Bendahan D, Mattéi JP, Fabreguettes C, Vague P, Cozzone PJ. Reduced metabolic efficiency of skeletal muscle energetics in hyperthyroid patients evidenced quantitatively by in vivo phosphorus-31 magnetic resonance spectroscopy. Metabolism. 1998;47:769-776.
5. American Association of Clinical Endocrinologists. Medical guidelines for clinical practice for the evaluation and treatment of hyperthyroidism and hypothyroidism. Endocr Pract. 2002;8(6):457-469.
6. Franklyn JA. The management of hyperthyroidism. N Engl J Med. 1994;33:1731-1738.
7. Starr M. Hypothyroidism Type 2: The Epidemic. Irvine, CA: New Voice Publications; 2010:177.8. Warmingham P. Effect of exogenous thyroid hormone intake on the interpretation of serum TSH test results. Thyroid Sci. 2010;5(7):1-6. http://www.thyroidscience.com/hypotheses/warmingham.2010/warmingham.7.18.10.pdf. Accessed June 30, 2011.9. Frost L, Vestergaard P, Mosekilde L. Hyperthyroidism and risk of atrial fibrillation or flutter: a population-based study. Arch Intern Med. 2004;164(15):1675-1678.
10. Woeber KA. Thyrotoxicosis and the heart. N Engl J Med. 1992;327(2):94-98.
11. Petersen P, Hansen JM. Stroke in thyrotoxicosis with atrial fibrillation. Stroke. 1988;19(1):15-18.
12. Sun L, Davies T, Blair HC, Abe E, Zaidi M. TSH and bone loss. Ann N Y Acad Sci. 2006;1068:309-318.
13. Benseñor IM, Lotufo PA, Menezes PR, Scazufca M. Subclinical hyperthyroidism and dementia: the Sao Paulo Ageing & Health Study (SPAH). BMC Public Health. 2010;10:e298. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2887825/?tool=pubmed. Accessed June 30, 2011.
14. Osman F, Gammage MD, Franklyn JA, et al. Hyperthyroidism and cardiovascular morbidity and mortality. Thyroid. 2002;12(6):483-487.
15. Tomer Y, Huber A. The etiology of autoimmune thyroid disease: a story of genes and environment. J Autoimmun. 2009;32:231-239.
16. Thyroid Disease Manager. Chapter 8. Akamizu T, Amino N, De Groot LJ. Hashimoto’s thyroiditis. http://www.thyroidmanager.org/Chapter8/8-frame.htm. Accessed April 3, 2011.
17. Lee SL, Griffing GT. Hashimoto thyroiditis. http://emedicine.medscape.com/article/120937-overview. Accessed April 9, 2011.
18. Ott J, Promberger R, Kober F, et al. Hashimoto’s thyroiditis affects symptom load and quality of life unrelated to hypothyroidism: a prospective case-control study in women undergoing thyroidectomy for benign goiter. Thyroid. 2011;21(2):161-167.
19. Padberg S, Heller K, Usadel KH, Schumm-Draeger PM. One-year prophylactic treatment of euthyroid Hashimoto’s thyroiditis patients with levothyroxine: is there a benefit? Thyroid. 2001;11(3):249-255.
20. Bastenie PA, Vanhaelst L, Golstein J, Smets P. Asymptomatic autoimmune thyroiditis and coronary heart-disease: cross-sectional and prospective studies. Lancet. 1977;2(8030):155-158.
21. Heinonen OP, Gordin A, Aho K, Punsar S, Pyörälä K, Puro K. Symptomless autoimmune thyroiditis in coronary heart disease. Lancet. 1972;1(7754):785-786.
22. Thyroid Center of Santa Monica. Guttler RB. Screening of family members of patients with Hashimoto’s thyroiditis. http://www.thyroid.com/screeninght.html. Accessed April 3, 2011.
23. Boelaert K. The association between serum TSH concentration and thyroid cancer. Endocr Relat Cancer. 2009;16(4):1065-1072.
24. Erden G, Barazi AO, Tezcan G, Yildirimkaya M. Biological variation and reference change values of TSH, free T3, and free T4 levels in serum of healthy Turkish individuals. Turk J Med Sci. 2008;38(2):153-158. http://journals.tubitak.gov.tr/medical/issues/sag-08-38-2/sag-38-2-10-0706-16.pdf. Accessed June 30, 2011.
25. Hollowell JG, Staehling NW, Flanders WD, et al. Serum TSH, T4 and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab. 2000;87:489-499.
26. Nikolai TF, Brosseau J, Kettrick MA, Roberts R, Beltaos E. Lymphocytic thyroiditis with spontaneously resolving hyperthyroidism (silent thyroiditis). Arch Intern Med. 1980;140(4):478-82.
27. Canaris GJ, Manowitz NR. The Colorado Thyroid Disease Prevalence Study. Arch Intern Med. 2000;160:526-534.