Mood Disorders: How Understanding Underlying Mechanisms Enhances Patient Outcomes
Nathan Horek, ND
There are a number of theories and models to describe the pathophysiological mechanisms underlying mood disorders. The biology is complex and varied, yet with stepping stones of concrete and comprehensible mechanisms. This can provide distinct targets for intervention. Our role as naturopathic doctors is to use this information as a broad template from which to understand the patient and discover the individualized path to homeostasis and wellness.
For a number of years, the view of depression was dominated by a monoamine “chemical imbalance” hypothesis, mainly involving serotonergic and noradrenergic neurotransmission. However, a single and distinct mechanism for this simplified explanation of a complex illness has not been discovered. Current published research paints a picture that the monoamines are only 1 small piece of a larger puzzle.1 Therefore, it is clear from research that mood disorders including depression should be “re-conceptualized” to include gross brain changes connected to stress and neuro-endo-immunological mechanisms.2
We know chronic illness is a combination of contributing factors interacting to create a physiological adaptation. Depression is a chronic disease initiated or perpetuated by a maladaptive neural, neuroendocrine, and immunological stress response. Four main areas of this process involve1:
- Cognitive processing
- Sympathetic activation and the hypothalamic-pituitary-adrenal (HPA) axis
- Neuroplasticity and neurogenesis
- Inflammation and Immune activation
Cognitive Processing
The ability to take in new information from the environment, make sense of it, and respond accordingly is essential for healthy mood and cognition. Essential neurobiological mechanisms of cognitive processing have been shown to be dysfunctional in mood disorders. This involves the pre-frontal cortex (PFC), limbic system and, most importantly, the connections between these 2 areas. The PFC and limbic system are in constant communication with each other, and their connections are in constant flux. The structure is dynamic, even in the adult brain. It is the disruption of this communication – like 1 family member refusing to listen and another shouting louder than can be heard – that disrupts overall cognitive processing contributing to anxiety, depression, and other mood disorders.
The PFC is the most evolved part of the brain, with particular importance for executive function: planning, decision-making, controlling impulses, and motivation. The PFC can be further divided into specialized areas of function which all play a vital role in emotional regulation and self-awareness. Deeper in the brain, the “feeling” limbic system is evolutionarily more ancient, and plays an important role in raw emotions and unconscious experience: excitement, fear, anxiety, memory, and desire. This is composed of 4 main regions: the hypothalamus, the amygdala, the hippocampus, and the cingulate cortex. Again, each serves a slightly different role in processing information and contributing to a response: the hypothalamus controls stress, the amygdala is responsible for extreme negative emotions such as fear and fright, the hippocampus controls the formation of long-term memories and calms the HPA, and the cingulate cortex plays a role in focus and attention.3
Environmental stressors to the human body are transmitted to the amygdala and cortex via the thalamus, activating the HPA system. Critical in mood disorders are the connections the amygdala has with regions of the PFC that play a role in emotional regulation and stress response inhibition, such as the orbitofrontal cortex (OFC), medial PFC, and anterior cingulate cortex (ACC). These areas then project back to limbic structures such as the amygdala and hypothalamus.1,4 Animal studies have indicated that these projections help control the amygdala and hypothalamus, modifying the stress response.5 In individuals with major depressive disorder (MDD), imaging studies have revealed a decreased volume of the ACC, decreased activity of the dorsolateral PFC, and hyperactivity of the amygdala.6,7 This imbalance reflects a hyperactive HPA with diminished regulatory capacity – a physical obstacle to cognitively processing external and internal information and adequately inhibiting a stress response. In addition, the hippocampus, responsible for cementing all new memories, has been demonstrated to have decreased volume and abnormal activity in depression.2 This data reflects the experience of depression: daily activities producing painful stimuli in the body. This is because the neural structures and connections necessary for emotional communication and the formation of new memories show structural and functional impairment. This is a physiological gap in mental and physical stress response management and observable pathology.
Sympathetic Activation & HPA axis
Activation of the HPA axis is initiated in limbic structures via direct and indirect projections to the hypothalamus, occurring without conscious awareness. A neuroendocrine cascade triggers the systemic release of glucocorticoids (cortisol), DHEA, norepinephrine, and epinephrine. Multiple negative feedback loops exist to prevent this train from running off the tracks. These are present in the hippocampus, hypothalamus, and anterior pituitary, and serve to inhibit this HPA cascade. For instance, cortisol acts on the hippocampus, which in turn inhibits the hypothalamus with GABAergic projections.1
Short-term HPA activation increases arousal, cognitive functioning, and gluconeogenesis via catecholamines and glucocorticoids. However, long-term activation of the HPA system will eventually dissolve the compensatory negative feedback loops, contributing to poor recovery and neuroendocrine imbalance. Globally, we may see disruptions in circadian rhythm that cause fatigue, insomnia, and immune dysfunction. Molecularly, we see decreased expression of glucocorticoid receptors in the hypothalamus and hippocampus, so much so that it has been declared “a key mechanism in the pathogenesis of depression” in the literature.8 This connection is so strong that Tafet and Nemeroff, in a 2015 review of the links between stress and depression, state that “an increasing body of evidence supports the association between chronic stress and depression at the molecular level, where hyperactivity of the HPA axis, with the consequent increase of cortisol, represents one of the most consistent findings in both syndromal mood and certain anxiety disorders.”1,9 In addition, the evidence suggests a “U-curve” of glucocorticoid activity; that is, “dysregulation of the HPA axis towards both hyper- or hypoactivity manifesting as a risk to mental wellbeing.”10 This is reflective of what we see clinically in the variability of both stress and mood patient-presentations and HPA biomarker testing. It is also reflective of the dynamic whole-system dysfunction that occurs in a conventional diagnosis.
As stated above, patients with depression have consistently displayed statistically significant diminished hippocampal volume, demonstrating this important piece in both the negative feedback loop and cycle of depression as a complex chronic illness.11 In addition, hippocampal volumes have been shown to have significant negative correlation with duration of major depression, suggesting worsening hippocampal function with worsening illness.12 The HPA axis, sympathetic activation, and subsequent dysfunction of negative feedback loops demonstrate a key piece of the complex puzzle of mood disorders, as well as key areas of intervention.
Neuroplasticity & Neurogenesis
Another area of research focus in cognitive function, further expanding the picture of mental health, is neurotrophic factors that contribute to not only brain development, but also neuroplasticity and neurogenesis in the adult brain. Brain-derived neurotrophic factor (BDNF) is the most well-known and well-researched neurotrophic factor. It stimulates cells in the hippocampus to proliferate and migrate to the left PFC, and is stored in platelets where it can facilitate neurogenesis in damaged tissues. Decreased expression of BDNF has been shown to contribute to depression, while upregulation of BDNF has demonstrated a role in clinical recovery.13 Clinical studies have found lower levels of BDNF in the serum of those diagnosed with MDD compared with healthy controls.14 Chronic levels of glucocorticoids have been shown to induce neuronal cell damage and atrophy, impair neurogenesis in the hippocampus, and impair neuroplasticity.15 This suggests that there is clear cross-talk between cortisol and BDNF, and this relationship is central to both the pathogenesis and symptomatology of depression and mood disorders.
Inflammation & Immune Activation
Numerous studies in psychoneuroimmunology have taught us that an inflammatory response is not only initiated by physical injury and infection, but may also be activated in both acute and chronic psychosocial stress. The earliest evidence of inflammation associated with major depression dates back to 1991.16 Since this time, “a vast literature has reproduced these findings, and meta-analyses have revealed that peripheral blood elevations in cytokines and acute-phase reactant(s) are some of the most reliable biomarkers of increased inflammation in depressed patients,”16 in particular, interleukin (IL)-1β, tumor necrosis factor (TNF)-α and C-reactive protein (CRP), respectively. It has also been shown that the increased inflammatory markers in depressed patients return to control levels with successful antidepressant treatment.16 Furthermore, those who fail to respond to treatment show increased inflammatory markers.16 It is also well documented that the medical administration of inflammatory cytokines such as interferon (IFN)-α induces depressive symptoms.16 Those with depression also exhibit a greater immunological sensitivity to stress; eg, the stress of public speaking has been shown to increase nuclear factor-κB (NF-κB).18 Clearly, immune dysfunction, including immune activation and inflammation, remains another piece of the complex puzzle of depression and other mood disorders.
Psychosocial stress will activate the sympathetic nervous system and HPA axis. Long-term, this will cause negative feedback loop disruption. We also know that psychosocial stress will activate the immune system in susceptible individuals. Another link in this cycle is that, “psychosocial stressors are well-established precipitants of episodes of major depression.”16,17 Although the impact of the sympathetic nervous system on the immune system is complex and varied, it has been shown that catecholamines will “activate their receptors on immune cells” and stimulate the release of proinflammatory cytokines via the NF-κB cascade.1,18 This occurs primarily on macrophages and monocytes in peripheral blood.18
Prolonged increase of proinflammatory cytokines in the brain results in a decrease in neurotrophins such as BDNF, reduced neuronal repair, decreased neurogenesis, and activation of glutamatergic neurotransmission, leading to apoptosis and oxidative stress.18 This diminishes the ability of the brain to take in new information, make new connections and strengthen existing ones, and makes it difficult for the individual to perform the daily tasks that rely on these cognitive abilities. This is especially true in relation to social and emotional processing. Figure 1 displays the relationship of these mechanisms in the cycle it creates. This cycle can be used as a potential diagram of depression, showing how the patient’s physiology becomes bound by these poorly-regulated compensatory mechanisms.
Teasing apart these pieces allows us to better understand the underlying causes of depression. This biological map of interdependent and functional nuances provides a general terrain from which to assess biomarkers and provide intervention to improve the dynamic ability of the human body to rebalance itself back to homeostasis.
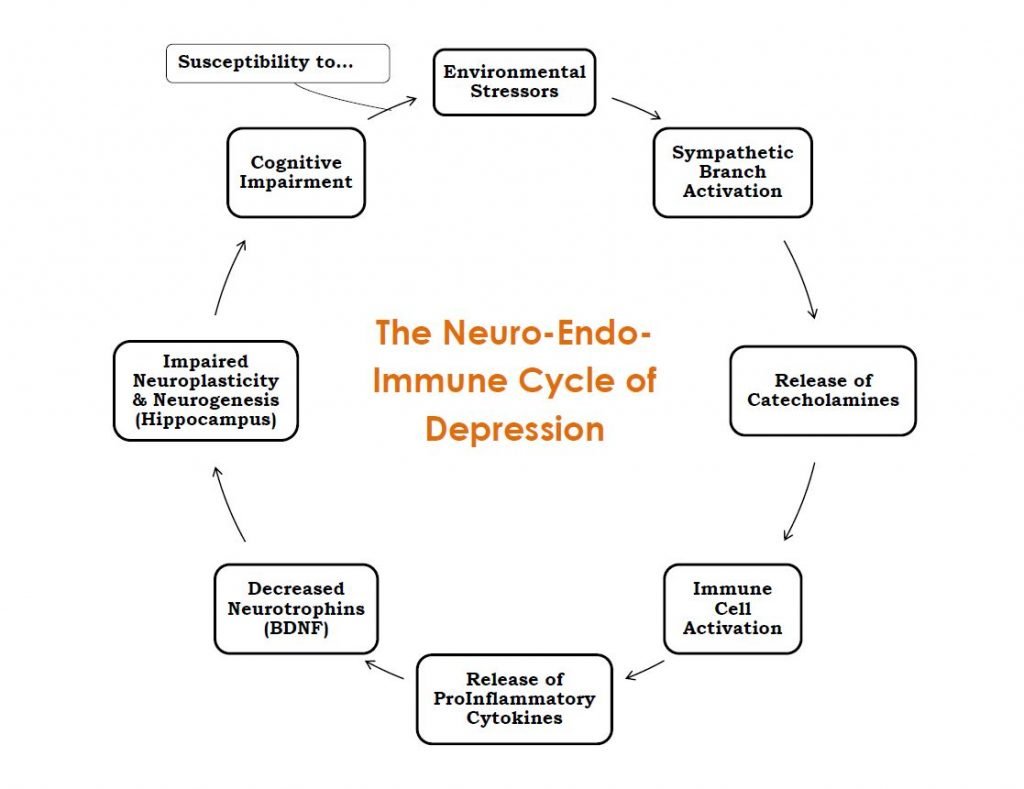
Figure 1. Neuro-Endo-Immune Cycle of Depression
Treatment Interventions
Since complex chronic diseases such as depression are a compilation of interrelated physiological mechanisms, addressing chronic disease is enhanced using the comprehensive and multi-modal approach of naturopathic medicine. While there is no 1 single cure for depression, a combination of interventions specific to individualized dysfunction provides a path for improvement and the best chance for recovery. This research is part of the road map from which this path can be blazed.
Exercise
Due to its central importance in brain health, a number of investigators have looked at ways to increase BDNF, primarily through exercise. Animal studies have shown an increase in both BDNF gene expression and protein levels in the hippocampus with a daily exercise regimen.19 This is exactly the effect we would want in order to induce to restore neurogenesis, neuroplasticity, and improve HPA regulation. Additional animal studies have shown that alternating days was as effective as daily exercise and that BDNF remained elevated for several days after exercise ceased, suggesting the positive effects to be more than transiently palliative. Furthermore, animals with prior exercise experience show a rapid return to normal BDNF levels upon only brief reintroduction to exercise.19 This suggests that exercise primes the body to make BDNF at the molecular level.
This has been confirmed in humans, and the BDNF increase seems to differ across exercise regimen, gender, and – for women – timing with menstrual cycle.20 A 2014 review of 32 studies concluded that BDNF is increased with a single session of aerobic exercise and further magnified with exercise frequency. The parameters of a “single session of aerobic exercise” ranged from 20 to 90 minutes at 75% of maximal heart rate.20 The “frequency” range was 45 minutes 3 times a week for 12 weeks, to 60 minutes 5 times a week for 6 months, at 50-75% heart-rate reserve.20 This is a reminder that setting specific parameters would prove more efficacious than a general recommendation. It also appears that these benefits are specific to aerobic exercise. In this same review, the authors state that a “majority of the studies suggested that strength training had no influence on peripheral BDNF.”20
Minerals
In addition to exercise, the micronutrients zinc, magnesium, and lithium have all demonstrated a relationship with BDNF and depression.21 In animal studies, zinc has been shown to enhance BDNF gene expression, while zinc deficiency has been associated with low BDNF in humans.21
Curcumin
Curcumin is a low-molecular-weight polyphenol in Curcuma longa that is widely considered inexpensive, orally bioavailable, and highly safe in humans. Research on curcumin and other curcuminoids is vast, with depression as a main focus point. The mechanisms for its actions in depression are thought to be related to monoaminergic activity, oxidative stress, HPA axis, and inflammatory pathways.22 More specifically, it has been shown to inhibit the monamine oxidase (MAO) enzyme responsible for the breakdown of serotonin, dopamine, and norephinephrine.23 Accordingly, it has also been shown to modulate the levels of these neurotransmitters essential to affective and cognitive function.23 In animal studies, curcumin has been shown to increase levels of BDNF and promote hippocampal neurogenesis.23
Where it is most well known and well researched is as an anti-inflammatory agent. Curcumin will inhibit the isoenzyme cyclooxygenase-2 (COX-2), NF-κB, and block the synthesis of the nitric oxide synthase enzyme.23 It has also been shown to lower the levels of IL-1β and block TNF-α, 2 cytokines essential to the immune dysfunction found in depression.23
Clinically, certain curcumin preparations have been shown to be as effective in depression as fluoxetine, with no adverse effects.22 It has been associated with greater improved outcomes on the Inventory of Depressive Symptomatology compared to placebo, and with greater efficacy in atypical depression.22 Patients treated with curcumin have also shown improvement at 6 weeks in both the Beck Depression Inventory (BDI-II) and the Hospital Anxiety and Depression Scale scores.23 Bioavailability among preparations remains controversial in the literature. Dosage seems to range around 1000 mg daily in these trials, and seems to require about 1 month for sustained mood improvement.
Omega-3 Fatty Acids
The literature has presented some contradictory results regarding the efficacy of omega-3 polyunsaturated fatty acids (PUFA) and fish oil for depression.24 This is probably due to the wide variability of study populations, dosage, ratio of eicosapentaenoic acid (EPA) to docosahexaenoic acid (DHA), and poor study-design (scientifically speaking, too many uncontrolled variables to support a healthy meta-analysis). Despite this, a 2014 comprehensive meta-analysis of randomized clinical trials found significant clinical benefit of PUFA treatment compared to placebo for patients with diagnosis of MDD, and for depressive patients without this diagnosis.25 A 2011 meta-analysis of randomized controlled trials, involving a total of 916 participants with depressive disorder or bipolar disorder, found that fish oil supplements containing >60% EPA produced benefit (as measured by standardized mean depression scores) versus supplements with <60% EPA, suggesting a stronger role for EPA than DHA in depression and bipolar disorder.26
Elevated glucocorticoids have been linked to both increased glutamatergic neurotransmission and decreased gray matter in those with depression. PUFAs have been shown to help protect glutamatergic neurotransmission from hyperactivation induced by stress and glucocorticoids.27 This presents a likely point for preventing depression onset in the face of stressful stimuli. In addition, animal studies have demonstrated increased serotonergic neurotransmission in the hippocampus when supplemented with fish oil.27
Mindfulness-Based Therapy
Mindfulness and meditation have been extensively studied across populations and conditions. In summary, mindfulness has been linked to outcome improvements, in terms of symptom reduction, biological markers, and neuroplasticity.28 The primary outcomes in which improvements are reported are in decreasing depression, anxiety, pain, and illness-related distress.28 One particular meta-analytic review found effectiveness for mindfulness-based therapy (MBT) across a number of conditions.29 In this review, MBT significantly reduced anxiety symptoms among those with an anxiety disorder, and was also effective for reducing depressive symptoms in those with a diagnosis of depression.29 The effect size was considered large for both of these instances. Two brain areas essential to focus, self-awareness, and emotional regulation – the dorsal medial prefrontal cortex (dmPFC) and anterior cingulate cortex (ACC) – have been shown to be activated during mindfulness meditation.30 In addition, structural MRI methods have produced a greater cortical thickness of the ACC in experienced meditators versus control subjects,30 the exact opposite pattern observed in those with depression.
HPA Axis Support
Another large area of research for depression prevention and treatment is lifestyle, nutritional, and botanical medicine for sympathetic activation and HPA-axis feedback dysfunction. For a well-written and well-researched review of neuroendocrine assessment and treatment of HPA disruption, see Dr Lise Alschuler’s “Optimizing the HPA Axis” in the August 2014 issue of NDNR.31
Basic Action Steps
This research creates an important foundation from which stem 3 important action steps: 1) understanding the complexity of depression and mood disorders as structural and metabolic chronic diseases involving multiple systems and pathways; 2) offering evidence-based first-line therapies for long-term outcome improvement; and 3) conceptualizing individualized treatment plans to reverse the course of the psycho-neuro-endo-immunological cascades of depression and other mood disorders.
 Nathan Horek, ND, received his doctoral degree in naturopathic medicine from the National College of Natural Medicine in 2011. He has a private practice in Minneapolis, MN, where he sees patients for complaints of mood, energy, sleep, digestion, and stress. Dr Horek feels passionately about using science to remove the stigma from mental health and increase patient access to non-pharmacological first-line therapies. With a BS in molecular biology, he enjoys doing research, writing, and teaching.
Nathan Horek, ND, received his doctoral degree in naturopathic medicine from the National College of Natural Medicine in 2011. He has a private practice in Minneapolis, MN, where he sees patients for complaints of mood, energy, sleep, digestion, and stress. Dr Horek feels passionately about using science to remove the stigma from mental health and increase patient access to non-pharmacological first-line therapies. With a BS in molecular biology, he enjoys doing research, writing, and teaching.
References
- Tafet GE, Nemeroff CB: The Links Between Stress and Depression: Psychoneuroendocrinological, Genetic and Environmental Interactions. J Neuropsychiatry Clin Neurosci. 2015;28(2):77-88.
- MacQueen G, Frodl T. The hippocampus in major depression: Evidence for the convergence of the bench and bedside in psychiatric research? Mol Psychiatry. 2011;16(3):252-264.
- Korb A. The Upward Spiral: Using Neuroscience to Reverse the Course of Depression, One Small Change at a Time. Oakland, CA: New Harbinger Publications, Inc; 2015.
- Ray RD, Zald DH. Anatomical insights into the interaction of emotion and cognition in the prefrontal cortex. Neurosci Biobehav Rev. 2012;36(1):479-501.
- Motzkin JC, Philippi CL, Wolf RC, et al. Ventromedial prefrontal cortex is critical for the regulation of amygdala activity in humans. Biol Psychiatry. 2015;77(3):276-284.
- Drevets WC, Savitz J, Trimble M. The subgenual anterior cingulate cortex in mood disorders. CNS Spectr. 2008;13(8):663-681.
- Zhong M, Wang X, Xiao J, et al. Amygdala hyperactivation and prefrontal hypoactivation in subjects with cognitive vulnerability to depression. Biol Psychol. 2011;88(2-3):233-242.
- Holsboer F. The corticosteroid receptor hypothesis of depression. Neuropsychopharmacology. 2000;23(5):477-501.
- Aborelius L, Owens MJ, Plotsky PM, Nemeroff CB. The role of corticotropin-releasing factor in depression and anxiety disorders. J Endocrinol. 1999;160(1):1-12.
- Baumeister D, LIghtman SL, Pariante CM. The interface of stress and the HPA axis in behavioural phenotypes of mental illness. Curr Top Behav Neurosci. 2014;18:13-24.
- Bremner JD, Narayan M, Anderson ER, et al. Hippocampal volume reduction in major depression. Am J Psychiatry. 2000;157(1):115-118.
- Colla M, Kronenberg G, Deuschle M, et al. Hippocampal volume reduction and HPA-system reduction activity in major depression. J Psychiatr Res. 2007;41(7):553-560.
- Duman RS, Monteggia LM. A neurotraophic model for stress-related mood disorders. Biol Psychiatry. 2006;59(12):1116-1127.
- Hashimoto K, Shimizu E, Iyo M. Critical role of brain-derived neurotrophic factor in mood disorders. Brain Res Brain Res Rev. 2004:45(2):104-114.
- Suri D, Vaidya VA. Glucocorticoid regulation of brain-derived neurotrophic factor: relevance to hippocampal structural and functional plasticity. Neuroscience. 2013;239:196-213.
- Haroon E, Raison CL, Miller AH. Psychoneuroimmunology meets neuropsychopharmacology: translational implications of the impact of inflammation on behavior. Neuropsychopharmacology. 2012;37(1):137-162.
- Kendler KS, Karkowski LM, Prescott CA. Causal relationship between stressful life events and the onset of major depression.Am J Psychiatry. 1999;156(6):837-841.
- Leonard BE. The concept of depression as a dysfunction of the immune system. Curr Immunol Rev. 2010;6(3):205-212.
- Berchtold NC, Chinn G, Chou M, et al. Exercise primes molecular memory for brain-derived neurotrophic factor protein induction in the rat hippocampus. Neuroscience. 2005;133(3):853-861.
- Huang T, Larsen KT, Ried-Larsen M, et al. The effects of physical activity and exercise on brain-derived neurotrophic factor in healthy humans: A review. Scand J Med Sci Sports. 2014;24(1):1-10.
- Greenblatt JM, Grossmann K. Mineral deficiencies and depression: Evidence-based research. In: Greenblatt JM, Brogan K, eds. Integrative Therapies for Depression. Boca Raton, FL: Taylor & Francis Group, LLC; 2016:115-132.
- Pentz JE. Botanicals for depression: Rhodiola, St. john’s wort, curcumin, and saffron. In: Greenblatt JM, Brogan K, eds. Integrative Therapies for Depression. Boca Raton, FL: Taylor & Francis Group, LLC; 2016:293-305.
- Kulkarni S, Dhir A, Akula KK. Potentials of curcumin as an antidepressant. ScientificWorldJournal. 2009;9:1233-1241.
- Pirec V, Brogan K. Integrative approaches to perinatal depression. In: Greenblatt JM, Brogan K, eds. Integrative Therapies for Depression. Boca Raton, FL: Taylor & Francis Group, LLC; 2016:421-440.
- Grosso G, Pajak A, Marventano S, et al. Role of omega-3 fatty acids in the treatment of depressive disorders: a comprehensive meta-analysis of randomized clinical trials. PLoS One. 2014;9(5):e96905.
- Sublette ME, Ellis SP, Geant AL, Mann JJ. Meta-analysis of the effects of eicosapentaenoic acid (EPA) in clinical trials in depression. J Clin Psychiatry. 2011;72(12):1577-1584.
- Lombard J. Genetic-based biomarkers in psychiatry: An integrative approach. In: Greenblatt JM, Brogan K, eds. Integrative Therapies for Depression. Boca Raton, FL: Taylor & Francis Group, LLC; 2016:241-249.
- Wolf C, Serpa JG. A Clinician’s Guide to Teaching Mindfulness: The Comprehensive Session-by-Session Program for Mental Health Professionals and Health Care Providers. Oakland, CA: New Harbinger Publications, Inc; 2015.
- Hofmann SG, Sawyer AT, Witt AA, Oh D. The effect of mindfulness-based therapy on anxiety and depression: a meta-analytic review. J Consult Clin Psychol. 2010;78(2):169-183.
- Grecucci A, Pappaianni E, Siugzdaite R, et al. Mindful emotion regulation: exploring the neurocognitive mechanisms behind mindfulness. Biomed Res Int. 2015;670724.
- Alschuler L. Optimizing the HPA axis. August 2014. Naturopathic Doctor News & Review. NDNR Web site. https://ndnr.com/endocrinology/optimizing-the-hpa-axis/. Accessed February 13, 2016.









